Conference 17, Case 2
Signalment:
2-wk-old domestic duck (Anas platyrhynchos var domestica)
History:
Conference 17, Case 2
Signalment:
2-wk-old domestic duck (Anas platyrhynchos var domestica)
History:
The duck was intranasally inoculated with Asian H5N1 subtype highly pathogenic avian influenza (HPAI) virus isolated in 2008. Cloudy eyes appeared on the third day after inoculation. The duck died on day 6 post inoc-ulation.
Gross Pathology:
Pancreatic focal necrosis and myocarditis were observed.
Laboratory Results:
N/A.
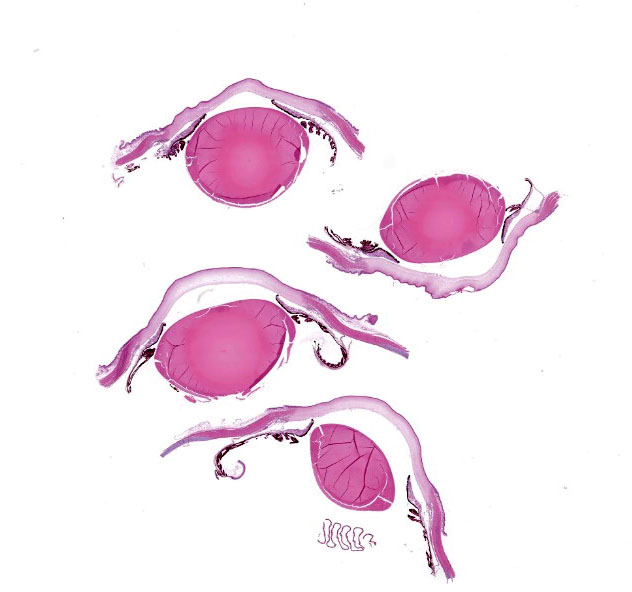
Microscopic Description:
The corneal stroma exhibited edema, scattered cellular debris, and inflammatory cell infil-trates that were predominantly located in the middle to deep layers of the stroma. Inflammatory cells were heterophils and foamy mac-rophages. Many corneal stromal spindle cells (keratocytes) became swollen. Corneal endo-thelium was diffusely lost, and the Descemet's membrane was exposed to the anterior cham-ber. Lymphoplasmacytic inflammation and focal loss of the muscular cells were observed in the iris. Inflammatory infiltrations were trapped in the pectinate ligament of the irido-corneal angle. Mild edema and inflammatory cells were observed in the conjunctiva.
Contributor’s Morphologic Diagnoses:
- Keratitis, heterophilic, with edema and diffuse corneal endothelial loss.
- Iritis.
Contributor’s Comment:
The H5Nx subtype HPAI virus is a global threat to the poultry industry and wild birds. The popular clinical signs of domestic ducks infected with the HPAI virus are depression, anorexia, decreased egg production, neuro-logic signs, and mortality. 9,10,17 In addition, corneal opacity (sometimes termed cloudy eye or blindness) is one of the characteristic and frequent findings in infected ducks. 1,6,9,13,18 Corneal ulceration and exophthalmos are rare findings in experimental infection.17 Ocular change by HPAI has been recorded in some waterfowl species, including bar-headed geese, cracking geese, herring gulls, laughing gulls, mallard ducks (including domesticated breeds), swan geese, and wood ducks.1-3,6,8,14,16
The corneal opacity is probably attributed to keratitis caused by the H5N1 subtype HPAI virus. The diffuse to focal loss of corneal en-dothelial cells was consistently observed in ducks inoculated with the virus.18 The corneal endothelium is a functional cell monolayer that maintains corneal transparency.15 The corneal endothelium transports water from the stroma to the anterior chamber and functions as a physical barrier to water movement.14 Alt-hough controversial, corneal endothelial cells are thought to have a limited capacity to re-generate.5,12,15 Therefore, an extensive loss of these cells can lead to corneal stromal edema and inflammation resulting in corneal opacity. Recovery from the opacity has been reported in geese and ducks experimentally infected with the H5N1 HPAI virus.8,18
The H5N1 HPAI virus can replicate in various types of ocular cells in domestic ducks. The type A influenza viral antigen can be detected by immunohistochemistry in the corneal en-dothelial cells for short period before its loss.18 The virus antigens can also be detected in the epithelial and stromal cells of the cornea, and cells of the iris, pectinate ligament, ciliary pro-cess, and pecten.18
Contributing Institution:
National Institute of Animal Health, NARO
3-1-5 Kannondai, Tsukuba, Ibaraki, Japan 3050856
JPC Diagnoses:
Eye (globe): Corneal endothelial and stromal necrosis, acute, diffuse, severe, with granulocytic and lymphocytic keratitis and anterior uveitis.
JPC Comment:
This case was an excellent example of how subtle lesions can clue a pathologist in to a specific etiology and how the evaluation of what is missing in a tissue is just as important as what is present. The lesions in this case were largely referrable to the necrosis and loss of corneal endothelium due to the HPAI infection. The corneal endothelium is responsible for removal of fluid from, and overall hydra-tion of, the cornea. Its absence contributed to marked corneal edema and subsequent stromal necrosis in this case. Stromal necrosis in the cornea can be a tricky diagnosis to make but is characterized by a lack of nuclei and slight tinctorial change to the stromal fi-bers of the cornea. These findings were most pronounced in the inner portions of the cornea (i.e., in the region closest to the corneal endothelium). While there are multiple causes of keratitis in Anseriformes (e.g., trauma, bacte-ria, fungi, WNV, immune-mediate disease), the specific loss of corneal endothelium ap-pears to be unique to HPAI.11
HPAI viruses primarily derive their virulence from the hemagglutinin (HA) protein, which effectively enables entry into host cells. HPAIs are usually of the H5 or H7 subtypes. Influenza A viruses also make use of, amongst many other virulence factors, non-structural protein 1 (NS1), which suppresses innate im-mune responses by blocking type 1 interferon production and signaling. NS1 does this by binding viral RNA and sequestering it away from cytosolic pattern recognition receptors, such as RIG1 and MDA5, effectively prevent-ing the downstream signaling pathways that would alert the cell to the presence of viral ge-netic material and subsequent interferon transcription.7 This process is crucial to enabling the virus to replicate in large numbers early in an infection and cause a high viremia. These viruses demonstrate marked tropism for endothelial cells and cause multifocal hemorrhage, edema, and necrosis. In birds, meningoencephalitis and pancreatic necrosis are the two most common lesions. Less commonly, the spleen, heart, and lungs are also affected. These lesions arise due to the vascular dam-age, thrombosis, and dysregulated immune re-sponses caused by the virus within the host, leading to tissue ischemia, necrosis, and, in severe cases, multiorgan failure.1,2,3
Conference participants also discussed the anatomy of the avian eye and how the lesions in this case were related to loss of corneal en-dothelial cells. Avian eyes have several unique anatomic features that are not present in their mammalian counterparts. The avian retina is avascular for enhanced visual acuity. Nutrition to the vitreous and retina is supplied by the pecten, which is a vascular plexus com-posed of small caliber blood vessels and pig-mented cells. Recent research demonstrates that the inner retina receives glucose from the pecten, which also removes lactic acid.4 Additionally, because of the pecten, trimming of an avian eye for histologic evaluation is slightly different than a mammalian eye. Rather than trimming the eye with a dorsoventral cut per-pendicular to the scleral arteries at the back of the eye as is standard for mammals, avian eyes require the cut to be made parallel with the arteries to ensure that the pecten is in section. The avian sclera often entombs cartilage and/or bone, which is particularly prominent in birds of prey. In these species, the scleral ossicles are so thick that the eyes require de-calcification prior to histologic processing. The iris also has various modifications depending on the species, perhaps the most strik-ing of which is in Strigiformes (owls). Owls have xanthophores that occupy the majority of the iridial stroma and impart the dramatic yel-low iridial coloration characteristic of many members of their species.11
References:
- Brown JD, Stallknecht DE, Beck JR, et Susceptibility of North American ducks and gulls to H5N1 highly pathogenic avian influenza viruses. Emerg Infect Dis. 2006;12(11):1663–1670.
- Brown JD, Stallknecht DE, Swayne DE. Experimental Infection of Swans and Geese with Highly Pathogenic Avian In-fluenza Virus (H5N1) of Asian Lineage. Emerg Infect Dis. 2008;14(1):136–142.
- Brown JD, Stallknecht DE, Swayne DE. Experimental infections of herring gulls (Larus argentatus) with H5N1 highly pathogenic avian influenza viruses by in-tranasal inoculation of virus and ingestion of virus-infected chicken meat. Avian Pathol. 2008;37(4):393–397.
- Damsgaard C, Skøtt MV, Williams CJA, et Oxygen-free metabolism in the bird inner retina supported by the pec-ten. Nature. 2026.
- Hirst LW, Bancroft J, Bi JQ, et Corneal endothelial response to induced myopia in the chicken. Clin Experiment Ophthalmol. 2001;29(4):244–247.
- Kishida N, Sakoda Y, Isoda N, et Path-ogenicity of H5 influenza viruses for ducks. Arch Virol. 2005;150(7):1383–1392.
- Mok BW, Liu H, Chen P, et The role of nuclear NS1 protein in highly pathogenic H5N1 influenza viruses. Microbes Infect. 2017;19(12):587-596.
- Nemeth NM, Brown JD, Stallknecht DE, Howerth EW, Newman SH, Swayne DE. Experimental infection of bar-headed geese (Anser indicus) and ruddy shelducks (Tadorna ferruginea) with a clade 2.3.2 H5N1 highly pathogenic avian influenza virus. Vet Pathol. 2013;50(6):961–970.
- Pantin-Jackwood MJ, Swayne Patho-genesis and pathobiology of avian influ-enza virus infection in birds. Rev Sci Tech.2009;28:113–136.
- Rhyoo MY, Lee KH, Moon OK, W et al. Analysis of signs and pathology of H5N1-infected ducks from the 2010-2011 Ko-rean highly pathogenic avian influenza outbreak suggests the influence of age and management practices on severity of dis-ease. Avian Pathol. 2015;44(3):175–181.
- Schmidt RE, Struthers JD, Phalen DN, eds. Special sense In: Pathology of Pet and Aviary Birds. 3rd ed. John Wiley & Sons, Inc; 2024:439-464.
- Schwartzkopff J, Bredow L, Mahlenbrey S, et al. Regeneration of corneal endothe-lium following complete endothelial cell loss in rat keratoplasty. Mol Vis.2010;16:2368–2375.
Spackman E, Pantin-Jackwood MJ, Lee SA, Prosser The pathogenesis of a 2022 North American highly pathogenic clade 2.3.4.4b H5N1 avian influenza virus in mallards (Anas platyrhynchos). Avian Pathol. 2023;52(3):219–228.
Sturm-Ramirez KM, Hulse-Post DJ, Go-vorkova EA, et Are ducks contributing to the endemicity of highly pathogenic H5N1 influenza virus in Asia? J Virol. 2005;79(17):11269–11279. - Tuft SJ, Coster DJ. The corneal endothe-lium. Eye. 1990;4(3):389–424.
- Yamamoto Y, Nakamura K, Yamada M, Mase Comparative pathology of chick-ens and domestic ducks experimentally in-fected with highly pathogenic avian influ-enza viruses (H5N1) isolated in Japan in 2007 and 2008. J Agric Res Q. 2010;44(1):73–80.
- Yamamoto Y, Nakamura K, Okamatsu M, et al. Avian influenza virus (H5N1) replication in feathers of domestic water-fowl. Emerg Infect Dis. 2008;14(1):149Yamamoto Y, Nakamura K, Yamada M, Mase M. Corneal opacity in domestic ducks experimentally infected with H5N1 highly pathogenic avian influenza virus. Vet Pathol. 2016;53(1):65–76.