CASE III:
Signalment:
14-month-old female intact common marmoset (Callithrix jacchus)
History:
This marmoset was presented with a sudden onset of lethargy and anorexia. The marmoset died approximately 18 hours post clinical evaluation and initiation of medical treatment.
Gross Pathology:
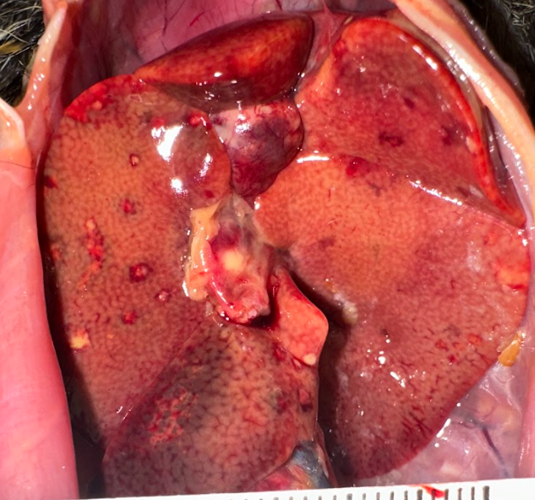
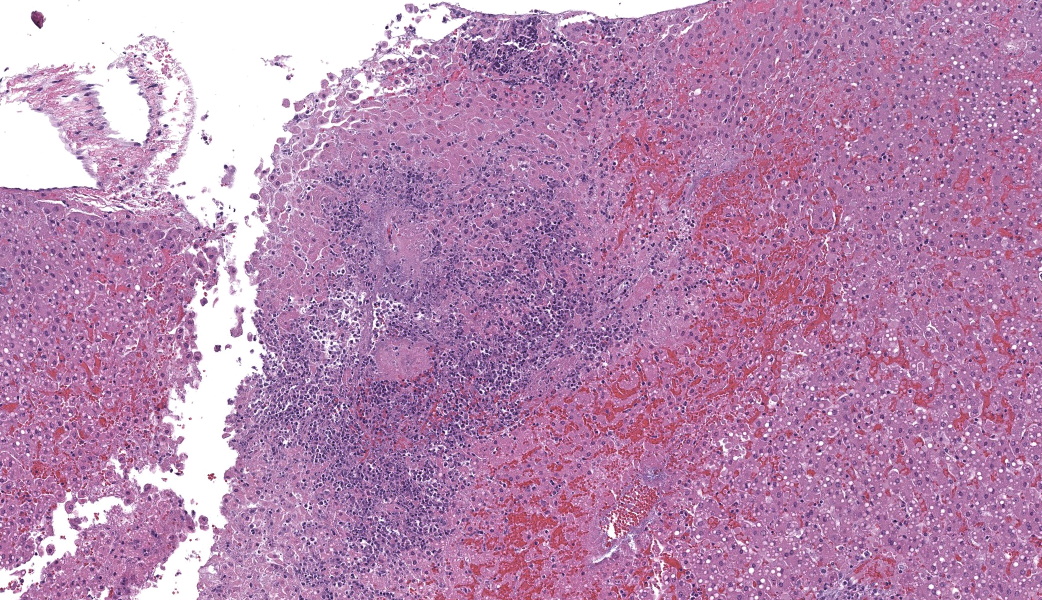
Examined was a female marmoset in fair nutritional condition (2.5/5) with mild post-mortem autolysis. Distributed throughout all liver lobes were yellow-white and red rimmed, ~0.5 cm diameter, randomly dispersed foci that extended into the parenchyma on cut section. The parenchyma was diffusely orange with an enhanced reticular pattern (Fig. 1). The lungs were diffusely bright red and atelectatic. Sections from all pulmonary lobes floated in formalin. There was marked diffuse gas dilation of the stomach. All segments of the small intestine were moderately to markedly distended by air and contained small amounts of digesta. The pancreatic lymph node adjacent to the duodenum was approximately 3x the size of normal. The cecum and colon had multifocal white to pale tan, circular areas of serosal discoloration. There were formed feces in the colon.
Laboratory Results:
Bacterial species identification confirmed by MALDI-TOF mass spectrometry:
Liver, aerobic culture: 3+ P. aeruginosa and <1+ K. pneumoniae
Liver, anaerobic culture: Clostridium barattii on thiglycolate
Lung, aerobic culture: 3+ P. aeruginosa, 1+ K. pneumonia and Ligilactobacillus rauteri
Cecum, aerobic culture: 3+ Mixed bacterial sps. included: Pseudomonas aeruginosa, Proteus mirabilis, Klebsiella pneumonia, Citrobacter freundii and Citrobacter farmer
- pneumonia string test: negative
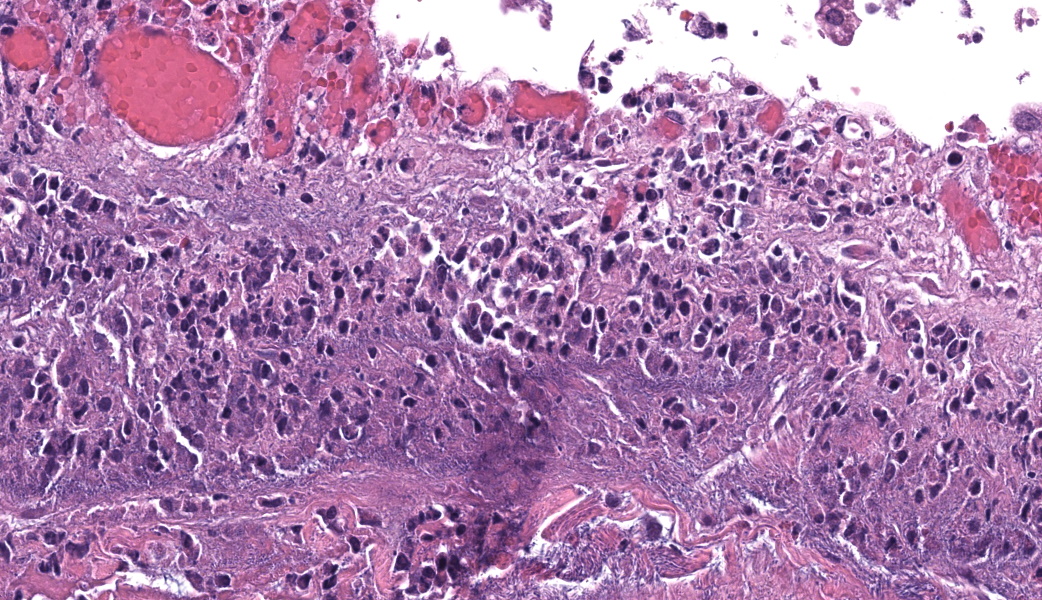
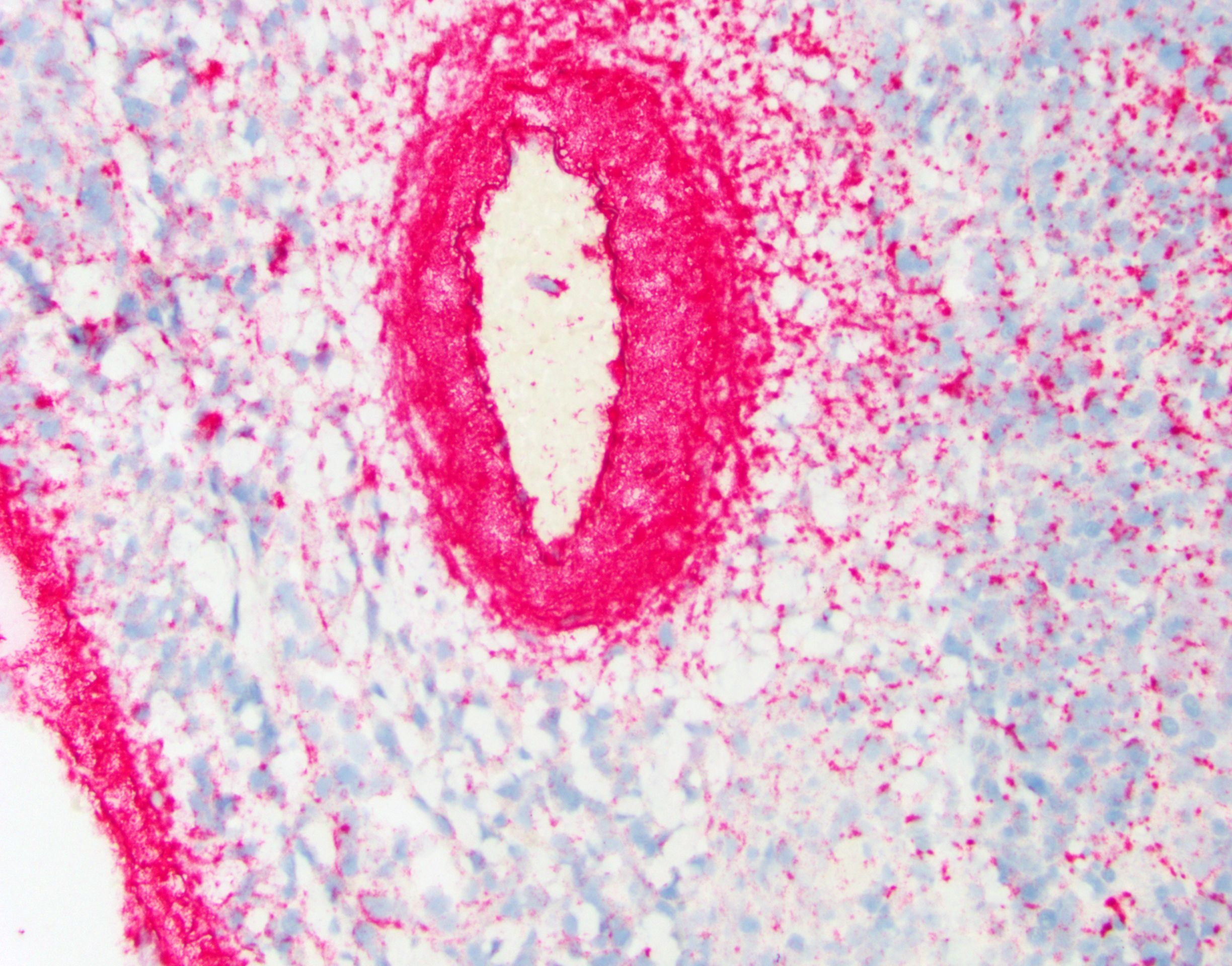
- aeruginosa ISH of liver: bacteria strongly positive, located diffusely within sinusoids and concentrated around vessels (Fig. 3 & 4).
Gram stain: Gram-negative rod-shaped bacteria associated with areas of inflammation and necrosis in the liver and gallbladder and presence of Gram-negative bacteria in circulation in the glomeruli and pulmonary vasculature.
Microscopic Description:
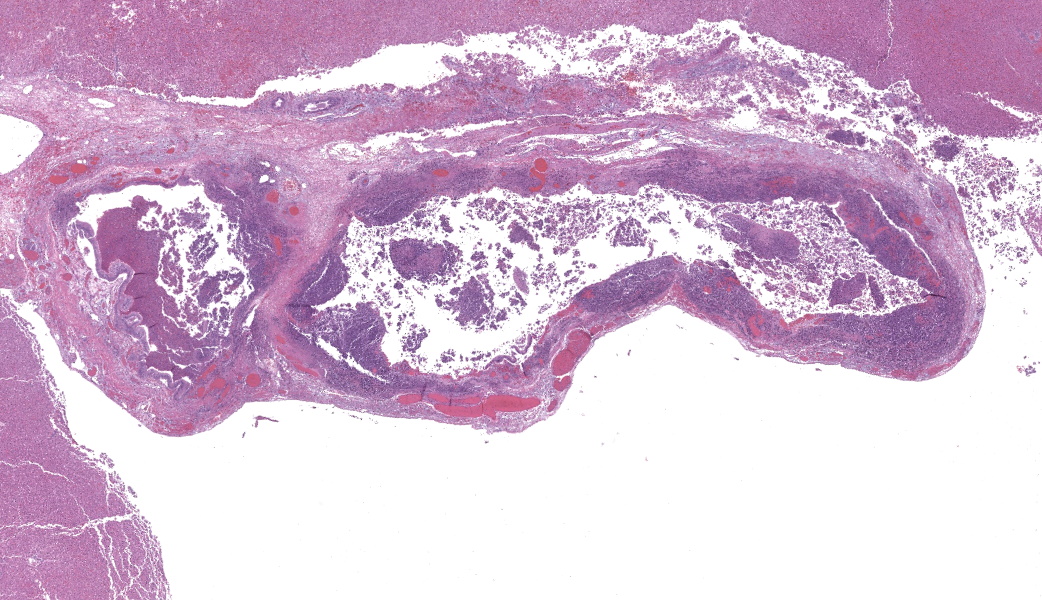
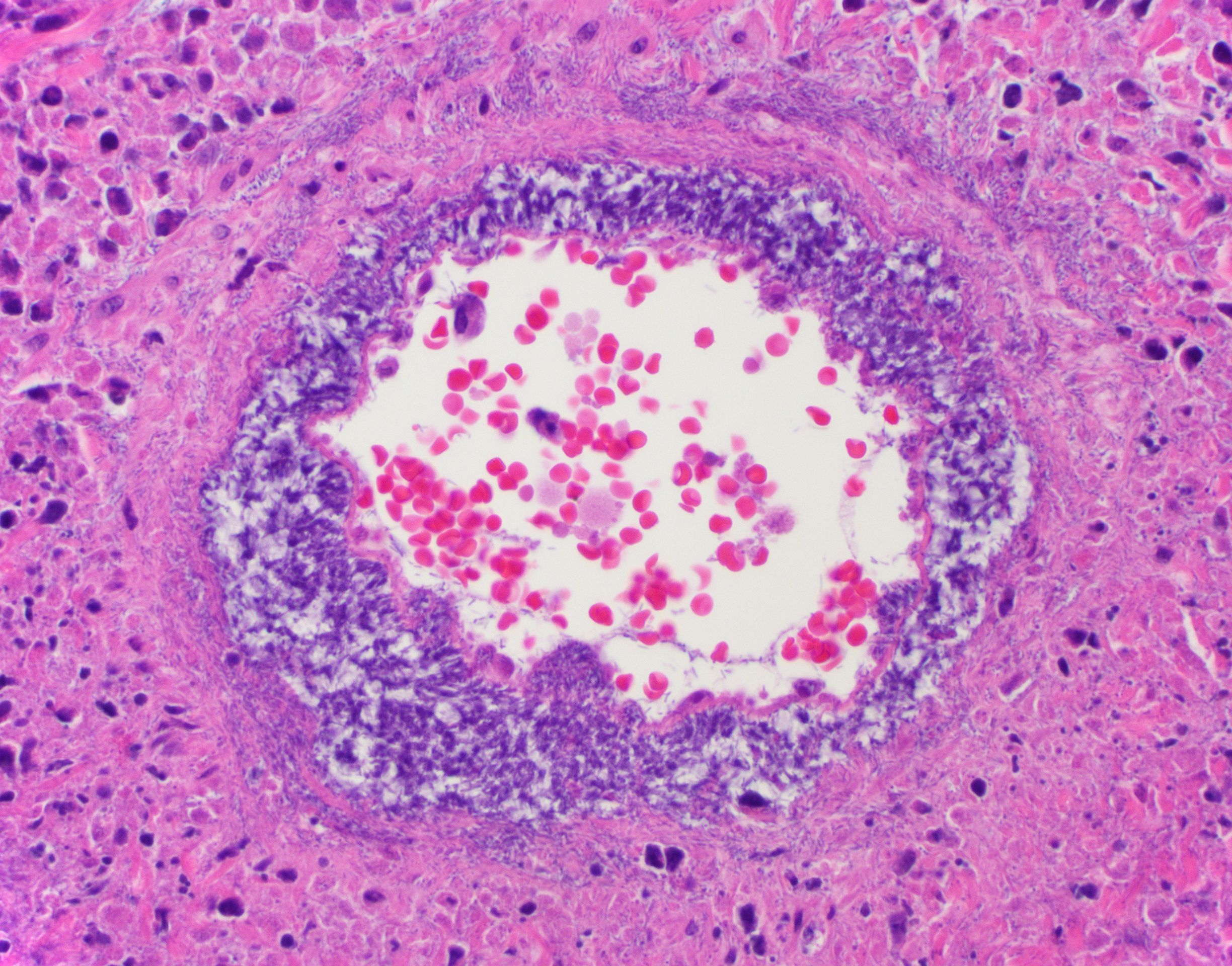
Gallbladder and liver: The gallbladder and adjacent portal tracts are effaced by multifocal to coalescing areas of coagulative and lytic necrosis admixed with foci of suppurative inflammation. The gallbladder is transmurally severely expanded by numerous neutrophils, scattered lymphocytes and histiocytes, abundant polymerized fibrin, necrotic debris eosinophilic proteinaceous fluid (edema), extravasated red blood cells (hemorrhage), and mats of extracellular rod-shaped bacteria (bacilli). Multifocally, blood vessels in the gallbladder are distorted by endothelial and/or mural necrosis and innumerable bacilli. Some vessels are partially occluded by fibrin and bacterial thrombi. Remaining intact vessels are congested. There is marked loss of gallbladder epithelium characterized by hypereosinophilia and loss of nuclear detail (necrosis), occasionally seen sloughing into the lumen admixed with neutrophils, fibrin, and red blood cells. Throughout the liver, bile ducts and portal vasculature undergo similar changes of varying severity (Fig 2). Bacilli are located extracellularly within portal areas and within sinusoids or intracellularly within macrophages. There are moderate, acute, multifocal loss of hepatocellular detail admixed with areas of fragmented chromatin and karyolytic debris (coagulative and lytic necrosis). Remaining hepatocytes are mildly to moderately swollen by small to medium-sized, round, clear, cytoplasmic vacuoles.
Contributor's Morphologic Diagnosis:
- Gallbladder: Severe, acute, multifocal to coalescing, necrotizing and suppurative, transmural cholecystitis with hemorrhages, congestion, polymerized fibrin, edema, and intralesional bacilli
- Vasculature, gallbladder and liver: Marked, acute, multifocal endothelial necrosis with mural bacterial colonization, bacteremia, and fibrin thrombi
- Liver: Severe, acute, multifocal, necrotizing and suppurative cholangiohepatitis with multifocal hemorrhages, acute random hepatic necrosis, and intralesional bacilli
- Liver: Mild to moderate, multifocal, microvesicular steatosis
Contributor's Comment:
The cause of death of this marmoset was due systemic bacterial infection that likely originated in the upper small intestine, resulting in an ascending biliary tree infection and translocation into the blood supply. Intravascular bacteria were identified in the heart, lung, kidneys, liver, gallbladder, duodenum, mesentery, cecum, and adrenal glands. Necrotic foci and hemorrhage were identified in the stomach, cecum, and mid-colon; these lesions lacked leukocytic cell infiltrates and were therefore considered peracute and secondary to sepsis/toxemia. Bacterial translocation across these lesions resulting in bacteremia cannot be ruled out.
In the case presented here, both P. aeruginosa and K. pneumoniae were isolated from the liver and lung. Given the gross and microscopic findings of acute hepatic necrosis and strong positive P. aeruginosa ISH, P. aeruginosa was determined to be the most likely primary pathogen. K. pneumoniae infections in non-human primates tend to form abscesses, which is not a feature of this infection.12 Additionally, we did not observe clear halos surrounding the bacilli when associated with leukocytes, which is a microscopic feature when dealing with hypervirulent K. pneumoniae outbreaks in NHPs. This marmoset also had a mild to moderate chronic lymphoplasmacytic enteritis with the duodenum most affected. Microbiome studies comparing healthy and sick captive marmosets affected by IBD or duodenal strictures/dilatation syndrome from different facilities demonstrated significant alterations in the diversity and shifts in their bacterial flora in the small intestine in sick marmosets.19,20 We cannot rule out the possibility that IBD may have contributed to alterations in the normal bacterial flora and facilitating the translocation of pathogenic bacteria in this case.
Pseudomonas species are motile, gram negative, bacillary, opportunistic pathogens of humans, animals, and plants. P. aeruginosa is associated with various diseases in veterinary species that include, but are not limited to, the following: septicemia in newly-hatched chickens and late stage embryos;11 mastitis in cattle, sheep, and goats;5,14 fleece rot in sheep;6 pyoderma, otitis externa, and ulcerative keratitis in in dogs and cats;8,17,18 hemorrhagic pneumonia in mink;1 metritis in mares;7 mouth rot (necrotic stomatitis) in snakes;4 otitis media, pneumonia, septicemia enteritis, and sudden death chinchillas;9 and wound infections in many species. In humans, this bacterium is often associated with infections in critically ill and immunocompromised patients. Over many years, P. aeruginosa isolates from hospital-acquired infections in humans have become increasingly antimicrobial resistant, and treatment options are limited.2
- aeruginosa contains an array of virulence factors, allowing it to cause a wide variety of clinical diseases.10 The outer membrane contains lipopolysaccharide, which modulates adherence to epithelial cells, evasion of phagocytosis, and activation of host immune response leading to dysregulated cytokine production. Biofilm formation is modulated by production of alginate and other exopolysaccharides. The flagellum allows for bacterial dispersion through aqueous and low-viscosity environments, relying on chemotaxis to swim toward epithelial cells. Pili are primarily used for epithelial cell adherence. A type III secretion system injects several cytotoxins and exotoxins that cause rapid host cell death. Several extracellular proteolytic enzymes, such as alkaline proteases and elastases, can cause significant host tissue damage and subsequent invasion. Because iron is required for bacterial growth, the bacteria has siderophores to scavenge this nutrient.10,22
- aeruginosahas been isolated from squirrel monkeys with meningitis, pododermatitis and cellulitis, and abscesses; an African green monkey with pneumonia and myocarditis; and from a chimpanzee with suppurative nephritis.21 Though P. aeruginosa is rarely a primary pathogen in marmosets a recent case series reported necrosuppurative cholangiohepatitis and necrotizing typhlitis in 7 indoor housed marmosets common.16 Infection was attributed to Pseudomonas contamination of the water supply. Though P. aeruginosa was frequently isolated from lesions in these cases, a subset of lesions recovered both E. coli and P. aeruginosa.16
Contributing Institution:
Laboratory of Comparative Pathology; Memorial Sloan Kettering Cancer Center, Weill Cornell Medicine, Hospital for Special Surgery, and The Rockefeller University 417 E. 68th St., ZRC-940 New York, NY 10065 Laboratory of Comparative Pathology
JPC Morphologic Diagnosis:
- Gallbladder: Cholecystitis, necrosuppurative, acute, diffuse, severe, with vasculitis and innumerable bacilli.
- Liver: Cholangiohepatitis, necrosuppurative, acute, multifocal to coalescing, severe, with portal vein thrombi, random hepatocellular necrosis, and innumerable bacilli.
JPC Comment:
This case, along with the outstanding contributor comment, provided an excellent opportunity to discuss necrotizing hepatitis in marmosets. Dr. Alves emphatically emphasized that necrotizing hepatitis in New World primates should immediately bring alphaherpesviruses to mind, including human herpes simplex virus (human alphaherpesvirus-1), macacine herpesvirus 1 (Herpes B), and saimariine herpesvirus 1 (formely known as Herpesvirus tamarinus or Herpes T).13 Other major differentials to consider include lymphocytic choriomeningitis virus (LCMV), and a wide array of "hot gram-negative bacteria" such as Francisella tularensis, Pseudomonas aeruginosa, and other fulminant septicemic organisms.13 To foot-stomp this point, participants received a firm reminder to know their alphaherpesviruses, as these agents remain among the most devastating and diagnostically urgent pathogens in Old and New World primates.13
The hepatic necrosis in this case was acute, and participants noted that cholestasis, which is generally considered a lesion of chronicity, was absent. The lack of viral inclusions also argued against many of the viral pathogens on the differential list. However, Dr. Alves cautioned against prematurely dismissing viruses simply because bacteria are abundant; many viral infections create the perfect environment for secondary bacterial invasion, and the presence of bacteria does not automatically identify the primary pathogen.
In this case, however, the evidence strongly supported Pseudomonas aeruginosa as the primary agent, with Klebsiella pneumoniae acting as a secondary invader. The bacteria are gram-negative and argyrophilic, and both were cultured from affected tissues. Klebsiella pneumoniae infections often produce overt abscessation, 'turkey egg" kidneys, and acute renal tubular necrosis.15 The absence of these lesions further supported the role of Klebsiella pneumoniae as a secondary pathogen, although it is more than capable of causing disease on its own.15
Participants discussed the most likely pathogenesis of infection as having ascended from the gastrointestinal tract, traveled up the common bile duct, and seeded the gallbladder and liver, producing the necrotizing cholangiohepatitis observed. This concurs with the contributor's proposed cause of death. The ascending route is well-documented in marmosets, particularly in the context of intestinal dysbiosis or chronic enteritis, and is a common route of infection for many enteric gram-negative bacterial pathogens.
Dr. Alves concluded the discussion with a concise but powerful review of the pathogenesis of gram-negative septicemia, emphasizing how rapidly this cascade can overwhelm animals. Circulating endotoxin (LPS) first binds to LPS-binding protein. This complex then engages CD14 on leukocytes. After dissociation of the binding protein, the CD14-LPS complex binds to TLR4, triggering a MyD88-dependent signaling cascade that drives NF-kB and interferon activation.3 The result is widespread macrophage stimulation with release of cytokines and nitric oxide, followed by endothelial activation and the induction of tissue factor. Tissue factor stimulates the extrinsic coagulation cascade. Once the coagulation cascade is initiated, endothelial injury accelerates, leading to consumptive coagulopathy and, ultimately, the risk of disseminated intravascular coagulation.3 This mechanistic walkthrough tied together the hepatic necrosis, vascular injury, and multiorgan involvement seen in the case, reinforcing why gram-negative septicemia remains such a devastating process.
References:
- Bai J, Wang X, Zhang Z, et al. Overview of Mink Immunity and Resistance to Pseudomonas aeruginosa. Vet Med Int. 2023;(1):6158844.
- Bassetti M, Vena A, Croxatto A, et al. How to manage Pseudomonas aeruginosa infections. Drugs in context. 2018;7.
- Bochud PY, Calandra T. Pathogenesis of sepsis: new concepts and implications for future treatment. BMJ. 2003;326(7383):262-266.
- Ciobotaru E, Tudor L, Constantinescu CM, et al. Pseudomonas spp. induced lesions in non-venomous snakes. J Comp Path. 2009;141(4):283.
- Dapgh AN, Hakim HS. Abouelhag HA, et al. Detection of virulence and multidrug resistance operons in Pseudomonas aeruginosa isolated from Egyptian Baladi sheep and goat. Vet World. 2019:12(10):1524.
- Denmann S, Tellam R, Vuocolo T, et al. Fleece rot and dermatophilosis (lumpy wool) in sheep: opportunities and challenges for new vaccines. Anim Prod Sci. 2021;62(4); 301-320.
- Ferrer MS, & Palomares R. Aerobic uterine isolates and antimicrobial susceptibility in mares with post-partum metritis. EVJ. 2018;50(2):202-207.
- Hillier A, Alcorn JR, Cole LK, et al. Pyoderma caused by Pseudomonas aeruginosa infection in dogs: 20 cases. Vet Derm. 2006;17(6);432-439.
- Hirakawa Y, Sasaki H, Kawamoto E, et al. Prevalence and analysis of Pseudomonas aeruginosa in chinchillas. BMC Vet Res. 2010;6:1-10.
- Jurado-Martin I, Sainz-Mejias M, & McClean S. Pseudomonas aeruginosa: an audacious pathogen with an adaptable arsenal of virulence factors. Int J Mol Sci. 2021;22(6);3128.
- Kebede F. Pseudomonas infection in chickens. J Vet Med Anim Health. 2010;2(4):55-58.
- Keesler RI, Colagross-Schouten A, Reader JR. Clinical and pathologic features of spontaneous Klebsiella pneumoniae infection in 9 rhesus macaques (Macaca mulatta). Comp Med. 2020;70(2):183-9.
- Matz-Rensing K, Bleyer M. Viral Diseases of Common Marmosets. The Common Marmoset in Captivity and Biomedical Research. 2019;251-264.
- Park H, Hong M, Hwang S, et al. Characterisation of Pseudomonas aeruginosa related to bovine mastitis. Acta Vet Hung. 2014;62(1):1-12.
- Pisharath HR, Cooper TK, Brice AK, et al. Septicemia and peritonitis in a colony of common marmosets (Callithrix jacchus) secondary to Klebsiella pneumoniae infection. Contemp Top Lab Anim Sci. 2005;44(1):35-37.
- Powers SJ, Castell N, Vistein R, et al. Bacterial Cholecystitis and Cholangiohepatitis in Common Marmosets (Callithrix jacchus). Comp Med. 2023;73(2):173-180.
- Pye C. Pseudomonas otitis externa in dogs. CVJ. 2018;59(11):1231.
- Santos TM, Ledbetter RC, Caixeta LS, et al. Isolation and characterization of two bacteriophages with strong in vitro antimicrobial activity against Pseudomonas aeruginosa isolated from dogs with ocular infections. Am J Vet Res. 2011;72(8):1079-1086.
- Sheh A, Artim SC, Burns MA, et al. Alterations in common marmoset gut microbiome associated with duodenal strictures. Sci Rep. 2022;12(1):5277.
- Sheh A, Artim SC, Burns MA, et al. Analysis of gut microbiome profiles in common marmosets (Callithrix jacchus) in health and intestinal disease. Sci Rep. 2022;12(1):4430.
- Simmons J, Gibson S. Bacterial and mycotic diseases of nonhuman primates. Nonhuman Primates in Biomedical Research. 2012:105-72.
- Rocha AJ, Barsottini MRDO, Rocha RR, et al. Pseudomonas aeruginosa: virulence factors and antibiotic resistance genes. Brazi Arch Biol Techn. 2019;62, e19180503.