Conference 17, Case 3
Signalment:
Adult carrion crow (Corvus corone) of un-known gender.
History:
An adult carrion crow was found dead in a humane trap. Only liver was submitted for examination.
Gross Pathology:
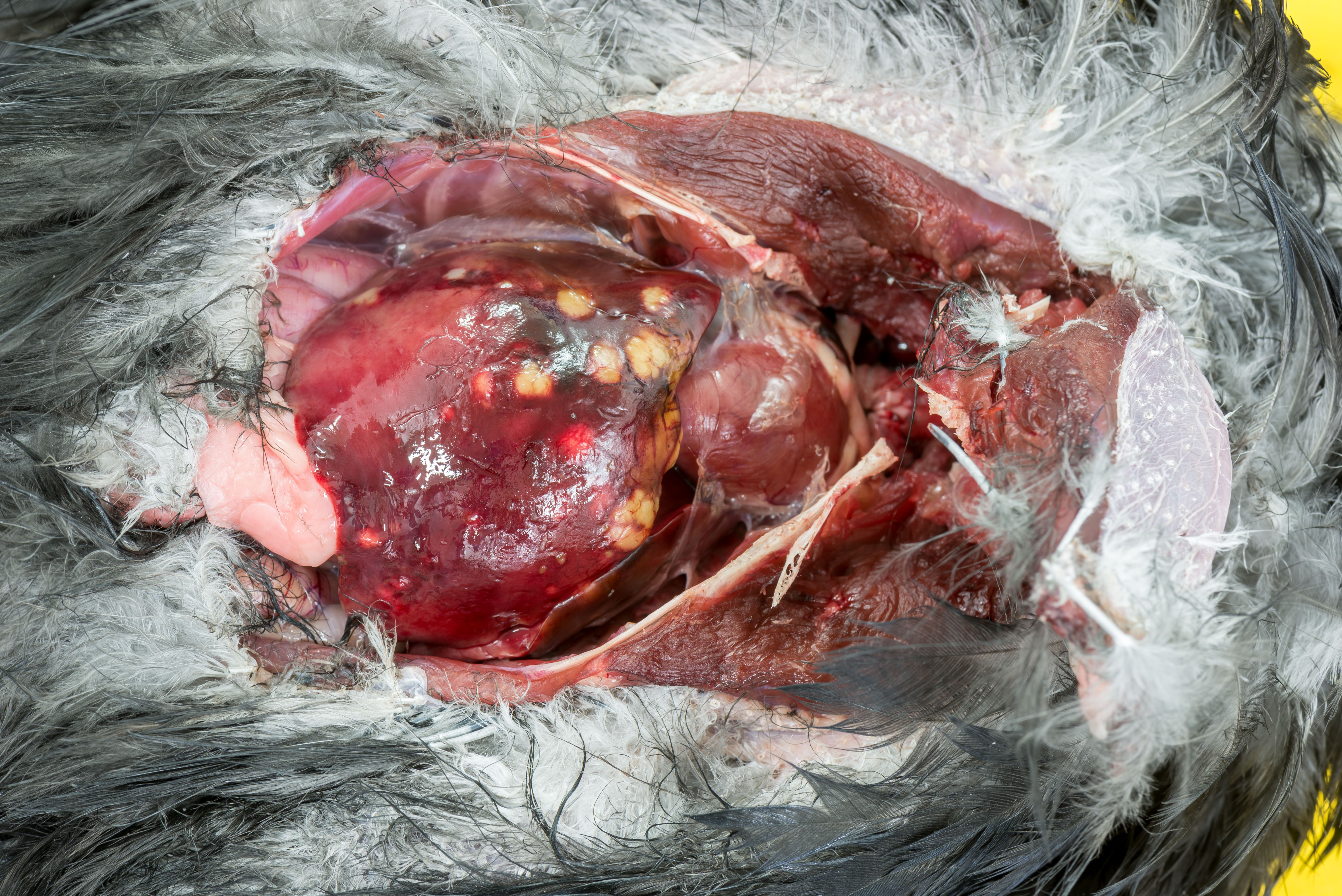
In a multifocal distribution, the liver contains numerous, cream/pale yellow, moderately firm, partially confluent nodules ranging from 1x1x1mm to 6x9x7mm.
Laboratory Results:
N/A
Microscopic Description:
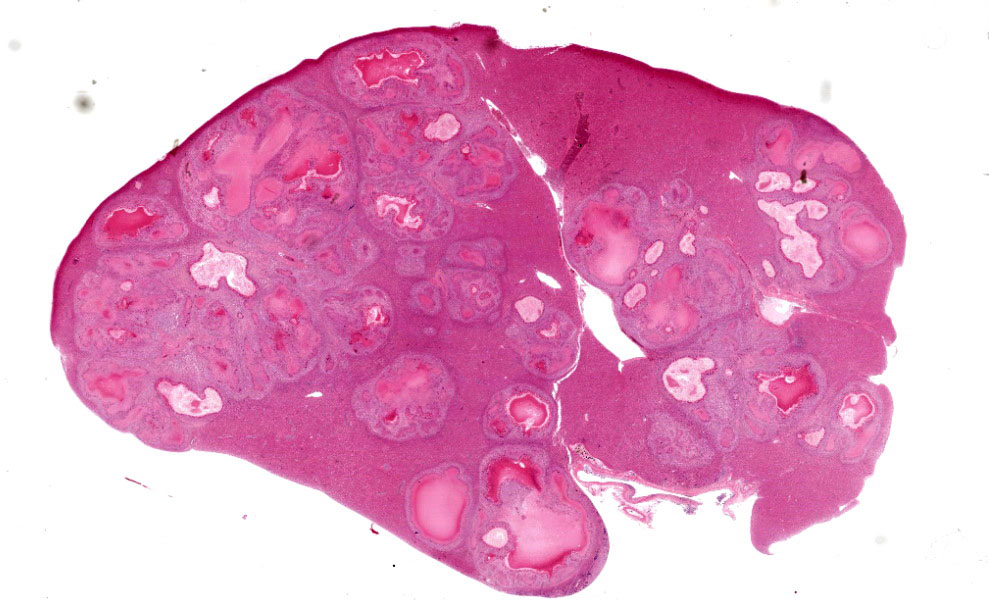
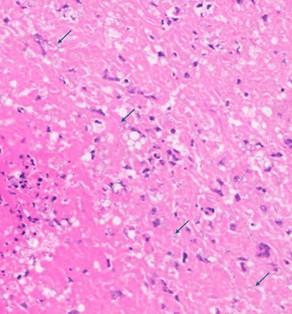
Approximately 70% of normal hepatic parenchyma is replaced by heterogeneously sized, multifocal to coalescing, randomly distributed foci (up to 5 mm in diameter) characterized by large, central, variably eosinophilic regions admixed with nuclear debris (necrosis), which are surrounded by inflammatory infiltrates (granulomas).
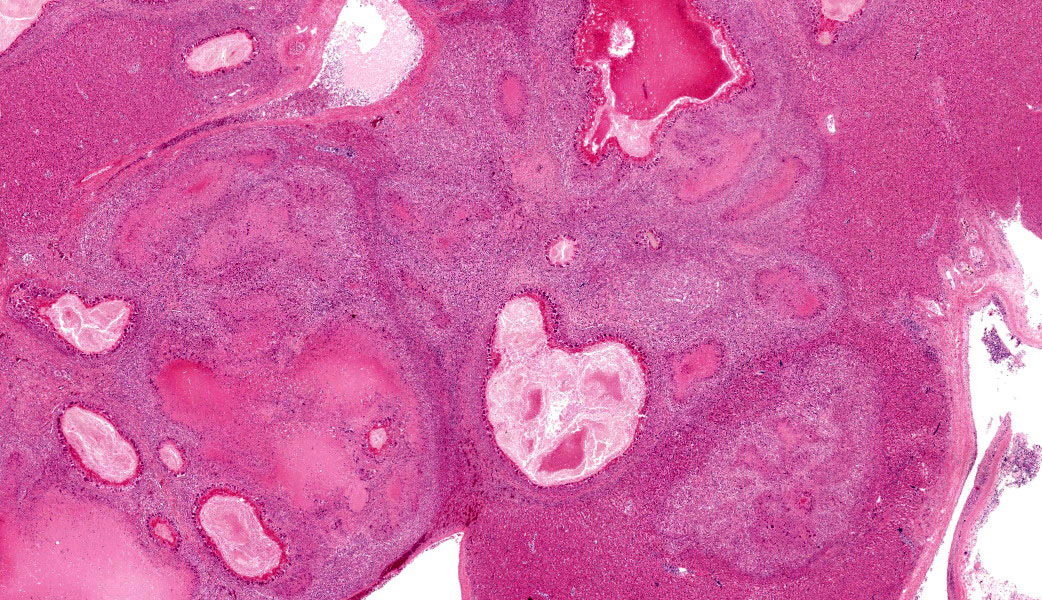
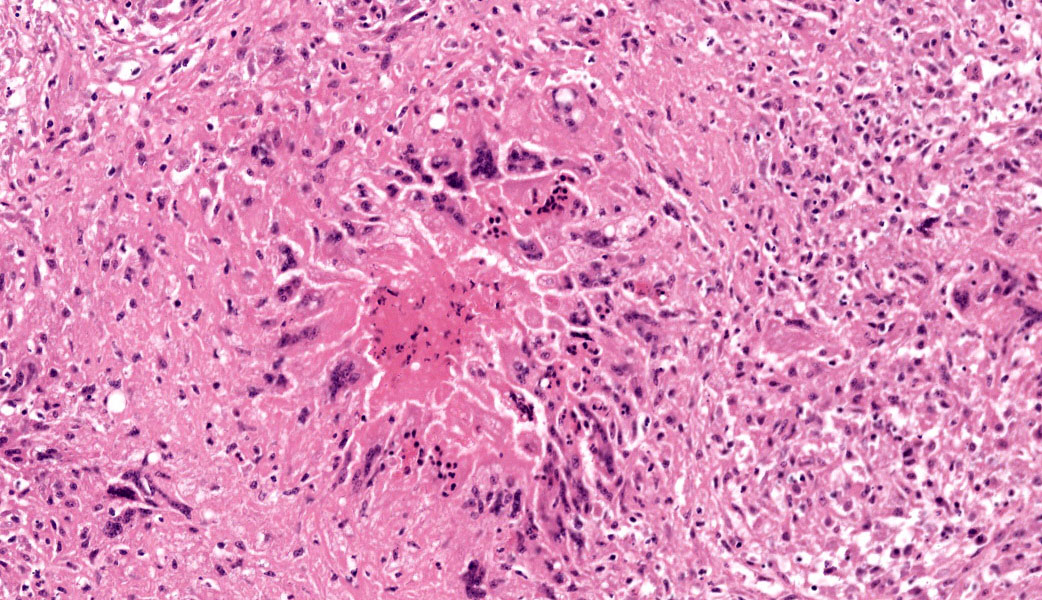
The inflammatory infiltrates consist of an inner, in most cases single layer of very large cells (up to 90 μm in diameter) with numerous (4-20) nuclei in a random arrangement (foreign body giant cells) abutting the area of necrosis, followed by a wider zone mostly comprising large (epithelioid) macrophages with occasional foreign body giant cells. Further peripherally moderate numbers of lymphocytes and plasma cells, and rare heterophils are present, multifocally situated within con-centrically arranged collagen with attendant spindle cells.
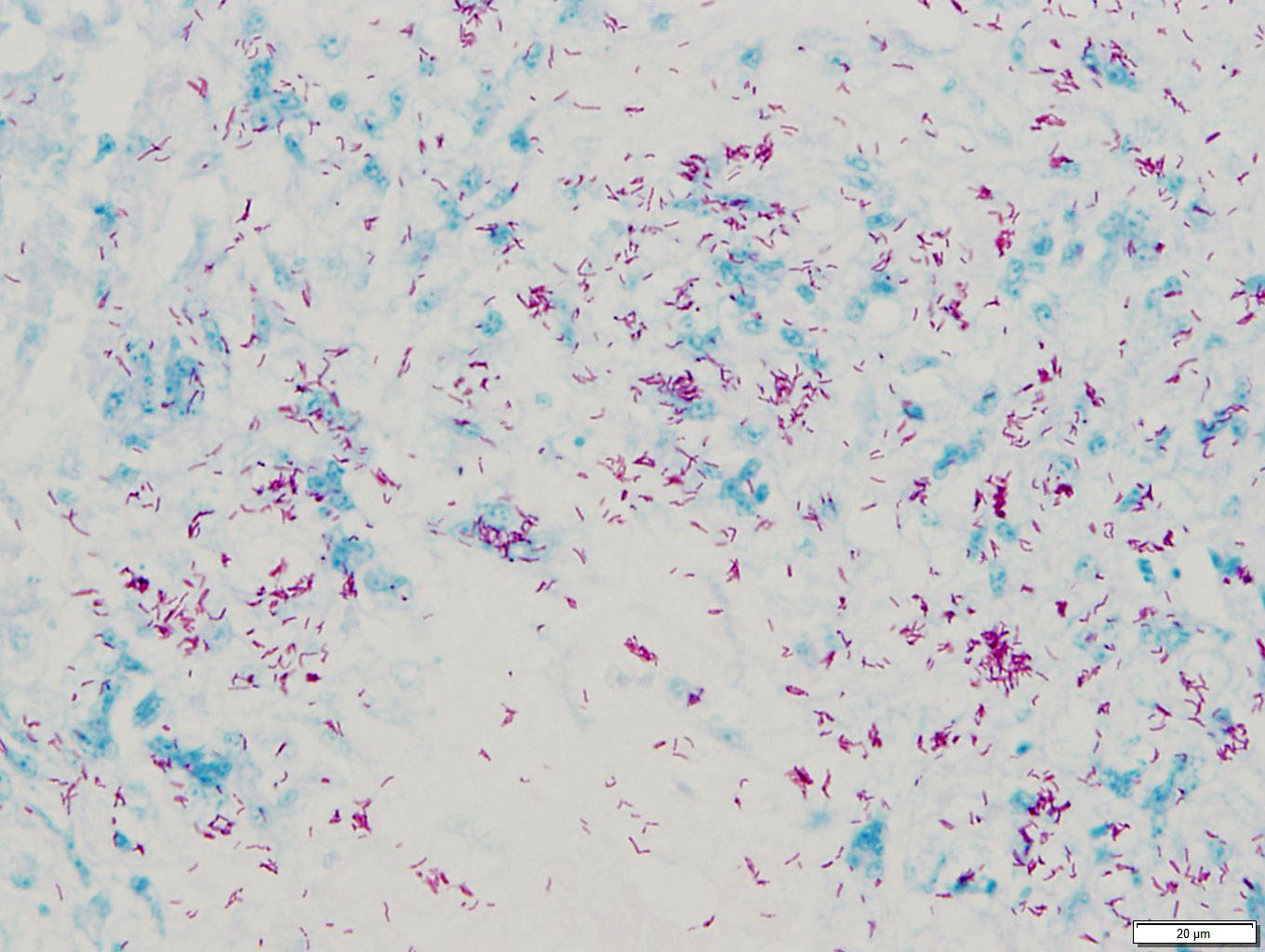
The areas of necrosis consist of variably in-tense eosinophilic amorphous central regions, which towards their periphery multifocally exhibit more distinct cellular outlines of homogenously eosinophilic (necrotic) cells and often small to moderately sized (10 – 30 μm) round clear spaces. Small numbers of granulo-mas contain short basophilic rods (length 3-5 μm) within the less densely arranged necrotic material. Additionally rare individual heterophils also are seen.
The intima of a large portal blood vessel which only contains a thin to moderately thick layer of smooth muscle tissue (interpreted as large vein) in a multifocal to coalescing distribution is moderately expanded by small num-bers of macrophages admixed with larger numbers of lymphocytes and plasma cells, and rare heterophils, whilst the lumen of this vessel contains moderate to large numbers of monocytes, erythrocytes and heterophils and small numbers of lymphocytes and plasma cells. Focally, the mural lymphoplasmacytic infiltrates are associated with a small, well-defined accumulation of macrophages.
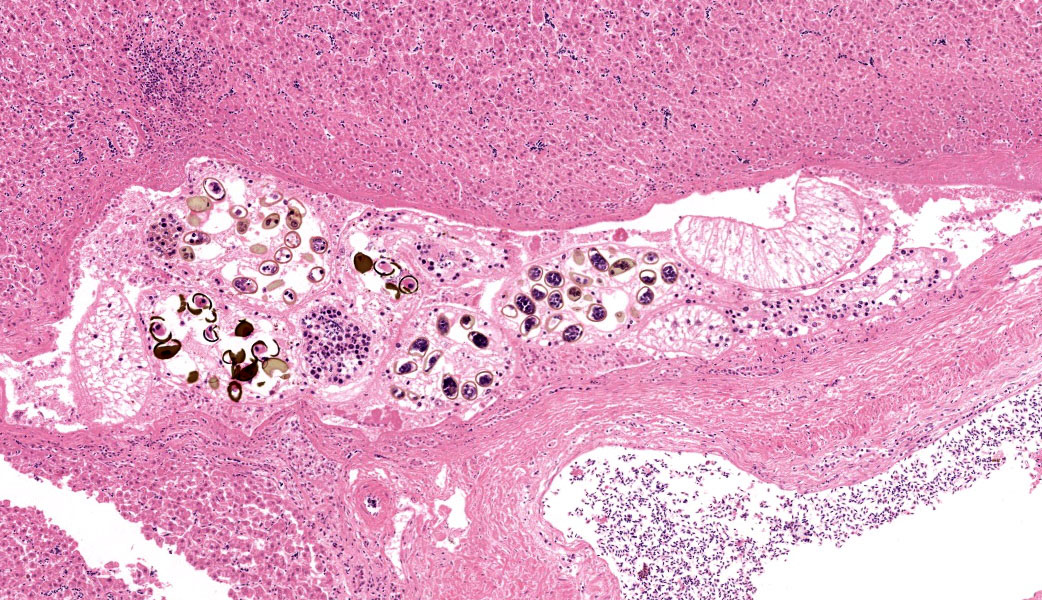
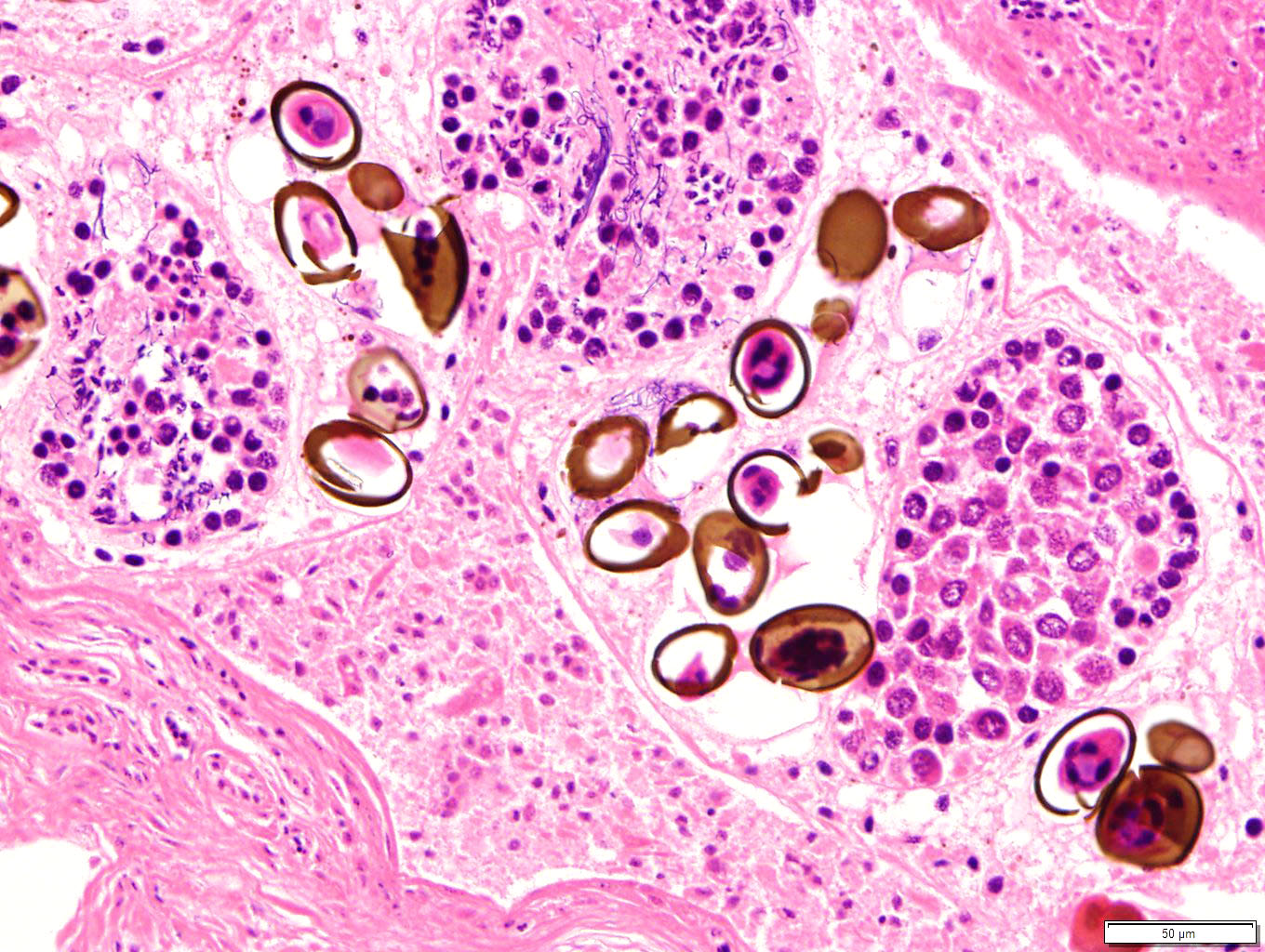
Additionally, the portal area described above contains small numbers of cavernous structures, which in small areas appears to be lined by cuboidal epithelium (interpreted as bile ducts) and contain small numbers of parasite profiles admixed with moderate numbers of desquamated epithelial cells and small numbers of macrophages and heterophils. The parasitic profiles are characterised by a thin (5-8μm) eosinophilic cuticle which lacks spines but occasionally appears ciliated, no apparent coelomic cavity, uteri containing numerous eggs and testes contain-ing linear basophilic spermatids within the same profile (hermaphroditic organism), vitellaria containing cells with homogeneous in-tensely eosinophilic cytoplasm, and a ventral sucker. The eggs, measuring approximately 20 x 45 μm, are oval although often unilaterally flattened (operculated), with yellow to brown 3-5 μm thick walls and contain a miracidium (trematode, consistent with Ly-perosomum longicauda). Small numbers of lymphocytes, plasma cells and heterophils are multifocally present in the wall of the bile ducts.
Contributor’s Morphologic Diagnoses:
Liver: Hepatitis, granulomatous, multifocal to coalescing, subacute to chronic, marked to severe with bacilli consistent with Mycobacterium spp.; and Phlebitis granulomatous, subacute to chronic, focal, moderate; and Cholangitis pyogranulomatous, multi-focal, chronic, minimal; with flukes consistent with Lyperosomum longicauda.
Contributor’s Comment:
Histological examination of the crow liver submitted revealed extensive effacement of the liver parenchyma by numerous, variably sized granulomas, many of which contain large numbers of acid-fast short rods. Additionally, focally a large vascular profile interpreted as a large vein exhibits a predominantly lymphoplasmacytic, focally also granuloma-tous intramural inflammatory infiltrate, whilst small and moderately sized cavities associated with the same portal area, devoid of erythrocytes and partially lined by cuboidal epithelium (interpreted as bile ducts) contain small numbers of trematode sections.
Considering the paucity of pathological changes associated with the flukes, these very likely need to be considered incidental findings in this case. The cause of the phlebitis is not entirely clear, however, taking the nature of the inflammatory infiltrate into account, this process may need to be considered to be associated with the mycobacterial infection either via local extension or possibly also via ex-tension from the lymphatics present within the vascular wall.
Avian hepatic granulomas may be of viral, bacterial, fungal, protozoal or helminth aetiology, with such agents exhibiting a preference for the liver or infecting the liver in an opportunistic fashion.13 Special stains revealed many of the hepatic granulomas in this bird to contain large numbers of short, acid-fast rods present both in macrophages and multinucle-ated giant cells and also extracellularly, which from their morphology and staining profile are most likely consistent with Mycobacterium spp.
Avian mycobacteriosis is primarily caused by M. avium (serotypes 1, 2, 3, and 6) and M. ge-navense, and is seen in found in many differ-ent birds including poultry, game, cage, wild and zoological species, predominantly of the North Temperate Zone. Less commonly M. intracellulare, M. scrofulaceum, M. fortuitum,tuberculosis and M. bovis are involved. The M. avium complex includes M. avium subsp. avium, M. avium subsp. paratuberculosis, M. avium subsp. silvaticum and M. intracellulare.5
Mycobacterium avium subsp. paratuberculosis, which causes Johne’s disease in rumi-nants, has both been isolated from tissue and feces of Scottish carrion crows (Corvus corone). Additionally, histological lesions consistent with M. avium subsp. paratuberculosis were observed in these animals, albeit described to be much milder when compared to those present in this crow.2 The infection of crows is surmised to be via the oral route, possibly from ingestion of infected rabbit or ruminant tissue. Experimentally, the contamination of livestock food by wildlife feces in turn has been determined as a possibility for the prevalence of paratuberculosis in the east of Scotland, and wild rabbits previously have been implicated as a possible wildlife reser-voir for paratuberculosis.4,12 Johne’s disease is relatively commonly diagnosed in cattle of the southwest of Scotland and an increase of this disease in small ruminants in Scotland also has been reported.10,11
Considering crows (in experimental conditions) have also been infected with M. bovis, however, here only the intraperitoneal inoculation, not the oral infection, resulted in myco-bacterial infection evident grossly, histologically or in pooled tissue cultures, M. bovis as a causative agent is considered less likely in this bird.3 Ultimately, PCR would need to con-ducted to be able to confidently identify the mycobacterial (sub-) species involved.
Generally, stress, age, immune status and possible pre-existing disease are considered pre-disposing factors for the infection of birds with Mycobacterium spp; transmission may be oral or via aerosols. Usually, granulomatous disease is present in multiple organs, including liver, spleen, pleura, intestinal serosa, bone marrow and lung, however, previous ex-amination of common crows with mycobacterial granulomas revealed a strong predisposition of the liver to lesions, with only rarely the spleen, peritoneum and lung affected.9,13 To what extent other organs were involved in the crow submitted unfortunately remains un-known. However, depending on the extent and distribution of other organs involved, the mycobacterial infection may have contributed to or even caused the death of this bird.
Dicrocoeliidae, which are small, lancet-like flukes occurring in the biliary and pancreatic ducts of vertebrates, represent a large family of trematodes with over 400 species. Their life cycle usually include two (snails and arthro-pods) or three (snails, arthropods and amphib-ians/reptiles) intermediate hosts. The genera and species present with this family are distin-guished from one another by the location and size of testes, ovary, vitellarium, suckers, cae-cum, genital pore, excretory vesicle and body form.
Lyperosomum longicauda of the genus Ly-perosomum is the common fluke of carrion crows (Corvus corone) in Europe. They ex-hibit a relatively elongate body with a thin cu-ticle lacking spines. The pharynx is large, whilst the oesophagus is short. Two caeca are present. The testes are close to round, orien-tated slightly oblique to one another and are found close the ventral sucker, which is larger than the oral sucker. The ovary is present cau-dal to the testes, whilst the genital pore is located just caudal to the pharynx. The vitellaria are relatively short and the uterus contains dark brown, thick-shelled eggs approx. 32 x 21 µm in size.7 Twenty species are present in the Lyperosomum genus, which may be found in intestines and livers of birds and mammals; the known life cycles include two hosts, namely terrestrial snails and insects.
Whilst most commonly trematodes in birds are present in the intestines, Lyperosomum longicauda is only found in the liver. This parasite also has been described in other birds including the song thrush (Turdus philomelos)
and the northern bald ibis (Geronticus er-emita).7 Canadian common crows (Corvus brachyrhynchos) exhibit Conspicuum macro-orchis of the genus Conspicuum, and Brachylecithum stunkardi of the genus Brachylecithum/ Lyperosomum within gall bladders and bile ducts.1
Contributing Institution:
Division of Pathology, Public Health and Dis-ease Investigation
School of Veterinary Medicine
College of Medical, Veterinary & Life Sciences
University of Glasgow 464 Bearsden road G61 1QH
Glasgow, Scotland, United Kingdom http://www.gla.ac.uk/schools/vet/
JPC Diagnoses:
- Liver: Hepatitis, granulomatous, chronic, multifocal and random, severe, with faintly staining bacilli.
- Liver: Adult trematodes, multiple.
- Liver, large-caliber veins: Phlebitis, lymphoplasmacytic, chronic, multifocal, moderate.
- Liver: Hepatitis, lymphoplasmacytic, chronic, multifocal and portal, mild.
JPC Comment:
This case was an excellent demonstration of mycobacteriosis in a bird. Many thanks to this contributor for providing this classic case. With careful scrutiny, the more recent granu-lomas contained numerous, faintly staining bacilli on H&E stain, which is typical of Mycobacterium spp. In many cases of paucibacil-lary mycobacteriosis, the bacilli can be seen at the center of “younger” granulomas, but they are characteristically poorly staining. This appearance can allow the pathologist to make a presumptive diagnosis of mycobacteriosis prior to confirmation with acid fast stains.
Paucibacillary mycobacteriosis is what is ex-pected in most cases of M. avium ssp avium tuberculosis, etc. due to these bacteria in-citing a Th1 response with resultant strong T-cell immunity.6 The stimulated Th1 cells pro-duce cytokines such as IL-1 and interferon to classically activate macrophages (M1 response). This enables the formation of granulomas to wall off the invaders and creates “tuberculoid” inflammation.6 Because the bacilli are predominantly seen only within the center of young granulomas rather than diffusely spread about, this form of mycobacteriosis is referred to as “paucibacillary.” On the other than, multibacillary mycobacteriosis is characterized by diffuse, disorganized inflammation with numerous bacteria that are poorly contained.6 This version of mycobacteriosis re-sults from Th2 activation and subsequent pro-duction of IL-4 and IL-13. These interleukins result in alternative (M2) macrophage activa-tion, resulting in “lepromatous” inflammation. M. avium ssp pseudotuberculosis (Johne’s dis-ease) and M. leprae (leprosy) are the classic examples of this type of mycobacteriosis, in which there are usually numerous acid-fast ba-cilli present throughout the inflamed tissue.6
Conference participants discussed at length whether the trematodes were present within a bile duct or within a portal vein. Support for portal vein includes: the shape of the luminal structure was similar to portal veins in the sur-rounding liver, the lumen contained erythrocytes (along with trematodes), the animal had substantial phlebitis, and definitive epithelial lining was not identified. Phlebitis was particularly pronounced in the large portal vein pro-file next to the trematode. Phlebitis is an un-common lesion of mycobacteriosis but can be seen in several other conditions in birds, including parasitism, aspergillosis, viral infection (e.g., WNV, HPAI, Paramyxovirus), sepsis, and (less commonly) immune mediated disease. Lead toxicity may also cause vasculitis, but it is typically fibrinoid vasculitis (unlikely the lymphocytic vasculitis seen in this case).12
Ultimately, participants were unable to definitively determine whether the trematodes were intravascular or within a bile duct. Immuno-histochemistry for pancytokeratin (AE1/AE3) was attempted to look for an epithelial lining to rule in/out a bile duct as the luminal structure. Unfortunately, IHC was non-contributory (internal controls were negative), likely due to autolysis in the specimen. Dr. Chris Gardiner was consulted on this case regarding the identity of the trematodes. Dr. Gardiner confirmed that this is trematode, specifically a hermaphroditic trematode with operculated eggs. The fact that this trematode is hermaphroditic at least ruled out schistosomes, as that particular genus of trematodes are gonochoric and do not have operculated eggs. Per Dr. Gar-diner, further determination of the trematode genus and species would require removal of whole specimens at necropsy and submission to a parasitologist.
References:
- Andrews SE, Threlfall Parasites of Common Crow (Corvus Brachyrhynchos)in I nsular Newfoundland. Proceedings of the Helminthological Society of Washing-ton. 1975;42:24-28.
- Beard PM, Daniels MJ, Henderson D, Pi-rie A, Rudge K, Buxton D, Rhind S, Greig A, Hutchings MR, McKendrick I, Steven-son K, Sharp JM. Paratuberculosis infection of nonruminant wildlife in J Clin Microbiol. 2001;39:1517-1521.
- Butler KL, Fitzgerald SD, Berry DE, Church SV, Reed WM, Kaneene JB. Experimental inoculation of European star-lings (Sturnus vulgaris) and American crows (Corvus brachyrhynchos) with My-cobacterium bovis. Avian Dis. 2001;45:709-718.
- Daniels MJ, Hutchings MR, Greig The risk of disease transmission to livestock posed by contamination of farm stored feed by wildlife excreta. Epidemiol Infect. 2003;130:561-568.
- Dhama K, Mahendran M, Tiwari R, Dayal Singh S, Kumar D, Singh S, Sawant PM: Tuberculosis in Birds: Insights into the Mycobacterium avium Vet Med Int. 2011;2011:712369.Frank KM and McAdam AJ. Infectious Disease. In: Robbins and Cotran, Pathologic Basis of Disease. 10th ed. Philadel-phia, PA: Elsevier; 2021:374.
- Gonenc B, Oge H, Oge S, Emir H, Ozbakis G, Asti C. First record of Ly-perosomum longicauda Rudolphi,1809 (Trematoda: Dicrocoeliidae) in Northern Bald Ibis (Geronticus eremita) in Turkey. Ankara Universitesi Veteriner Fakultesi Dergisi. 2012;59: 227-230.
- Mettrick Studies on the helminth par-asites of British birds. In: Department of Parasitology. University of London. 1957.
- Mitchell CA, Duthie RC. Tuberculosis of the Common Can J Comp Med Vet Sci. 1950;14:109-117.
- SAC C VS. Increase in the prevalence of Johne's disease in sheep in Scotland. Vet Rec. 2011;168:13-16.
- SAC S Johne's disease commonly diagnosed in cattle in south-west Scotland. Vet Rec. 2009;164:256-259.
- Schmidt RE, Struthers JD, Phalen Pa-thology of Pet and Aviary Birds. 3rd ed. John Wiley & Sons; 2024.
- Shaughnessy LJ, et High prevalence of paratuberculosis in rabbits is associated with difficulties in controlling the disease in cattle. Vet J. 2013;198:267-270.
- Supartika IK, Toussaint MJ, Gruys E. Avian hepatic A review. Vet Q. 2006;28:82-