Conference 19, Case 1:
Signalment:
Male, 408-gram, age unknown, Fischer 344 rat (Rattus norvegicus)
History:
The rat had telemetry placement surgery on 8 June 2015. Post-operative treatments included meloxicam, buprenex along with supplemental feeding. Animal recovered from surgery without complication and was acting normally during the initial post-surgery period. On the 26th of June, the rat appeared pale and lethargic and euthanasia was elected.
Gross Pathology:
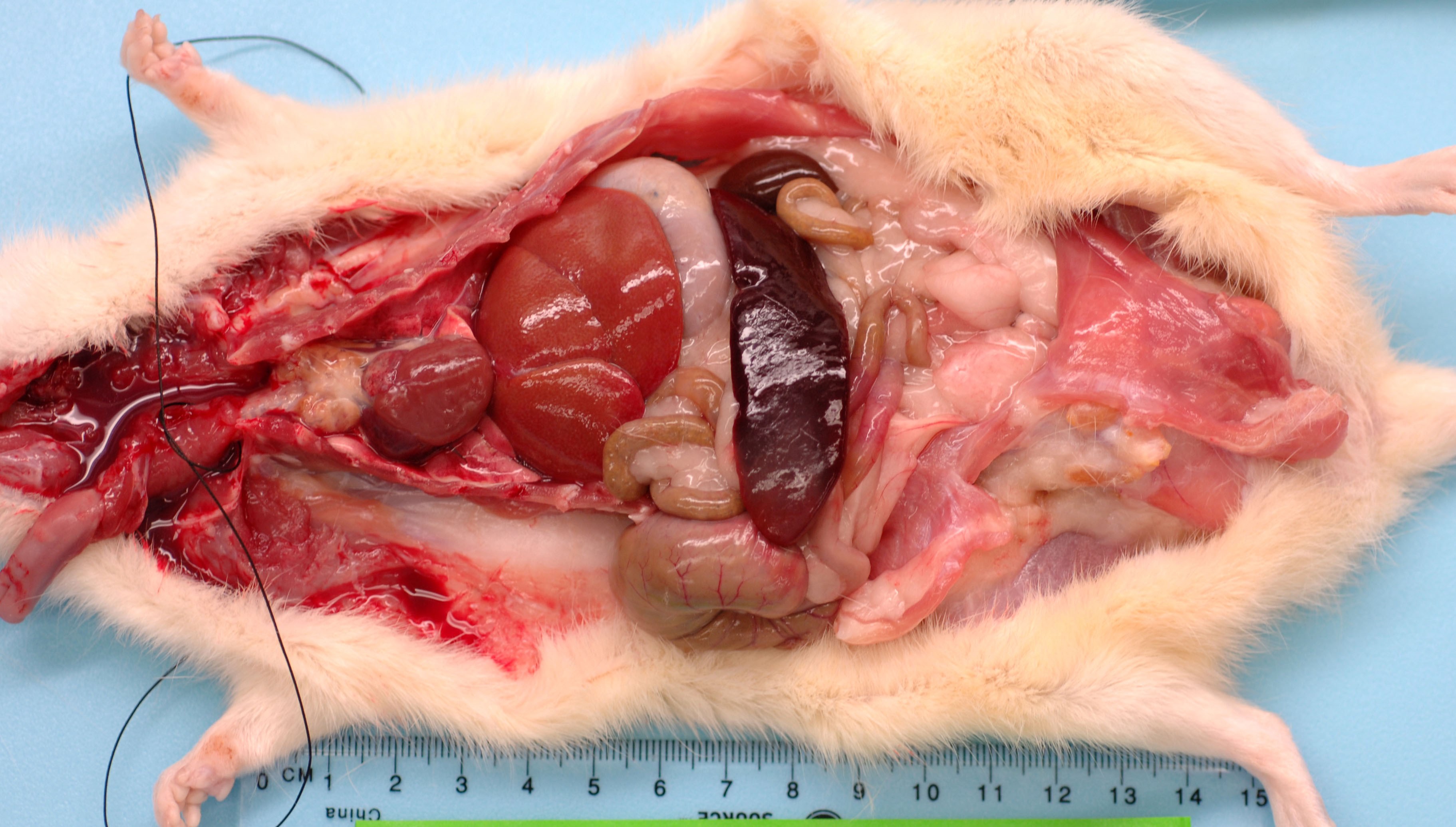
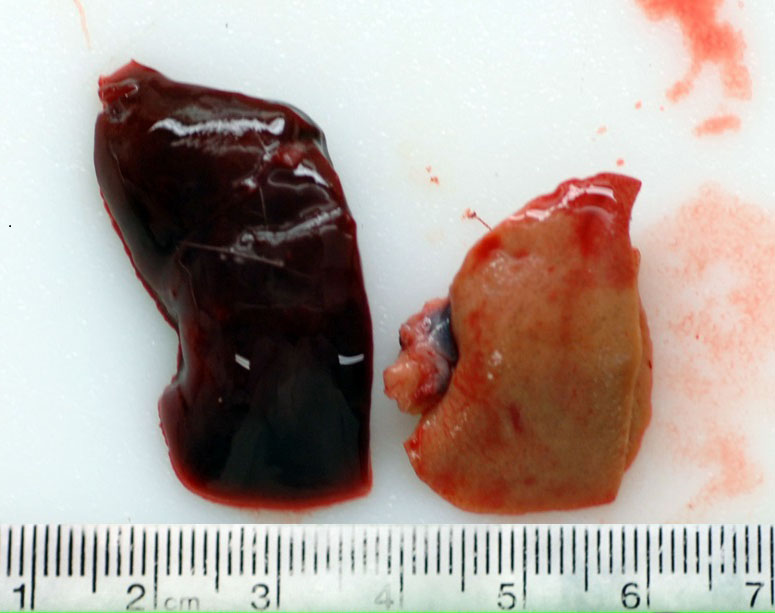
This rat has a body score of 3/5 with adequate subcutaneous and visceral fat. The skin incisions over the skull and left thorax from the telemetry surgery have completely healed. The pocket over the left thorax holding the telemetry device contains 5 ml of milky serosanguinous fluid. There is a layer of fibrin coating the telemetry device. The lungs were collapsed and diffusely red. The spleen is enlarged measuring 6.5 x 2 x 1 cm, friable with a homogenous pale red area on one pole (infarct). The liver is diffusely pale (light brown) and friable and the thymus is mottled tan/brown. The pancreas is soft and tan and both kidneys have a brown/green appearance. There is a moderate amount of dry food contents in the stomach, small amount of normal digesta and gas in the small intestine, moderate amount of soft green contents in the cecum and many dry fecal pellets in the colon. There is no urine in the bladder.
Laboratory Results:
N/A.
Microscopic Description:
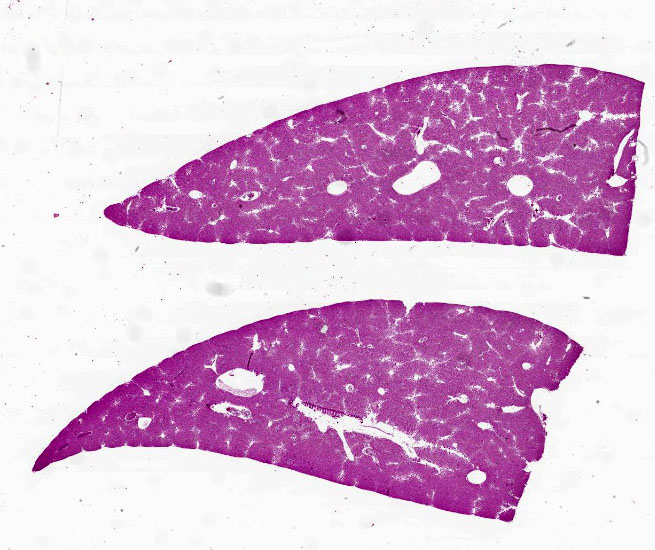
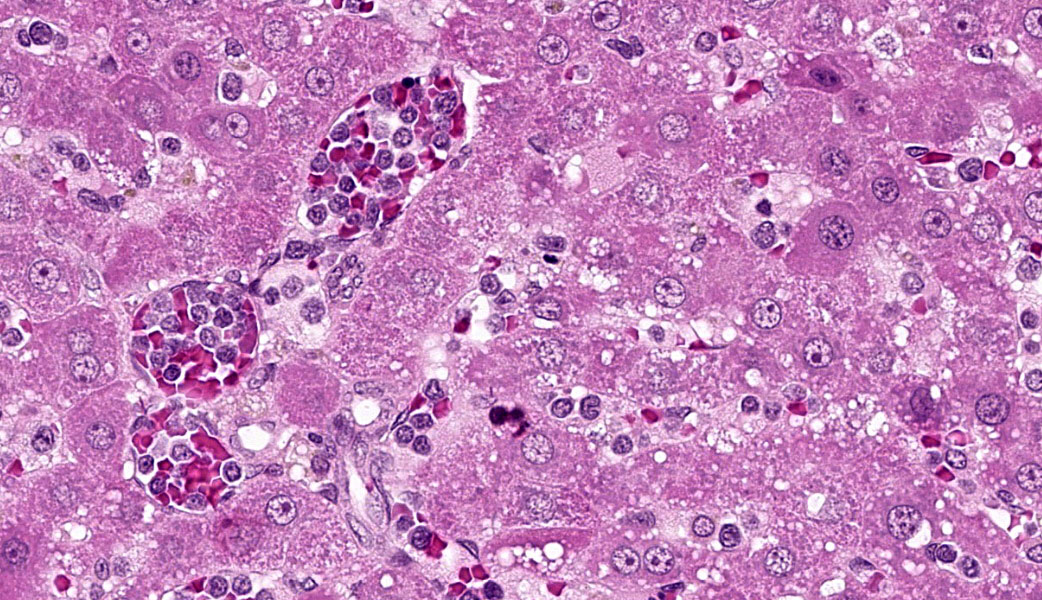
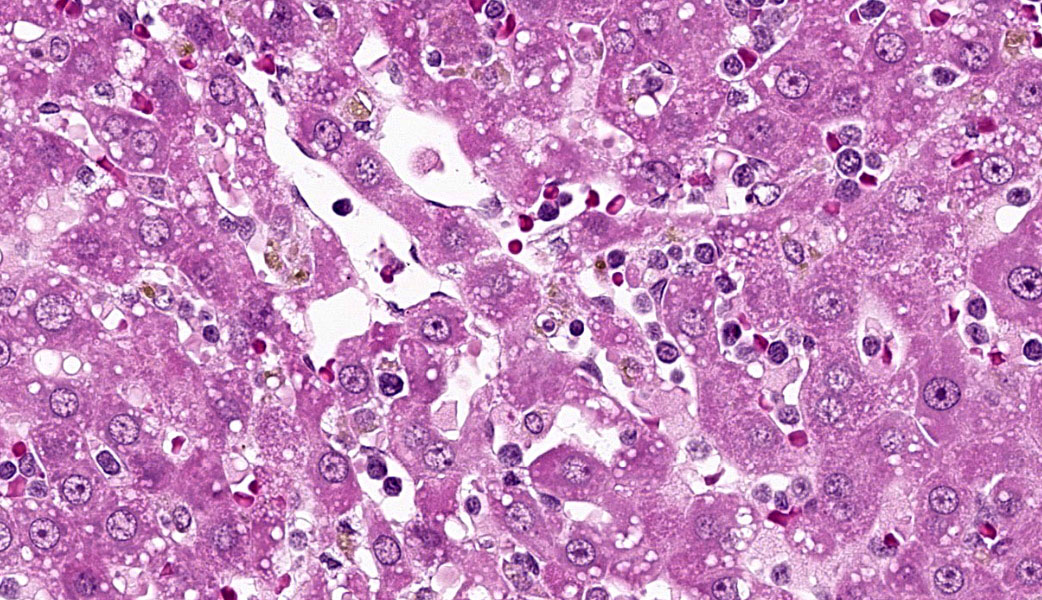
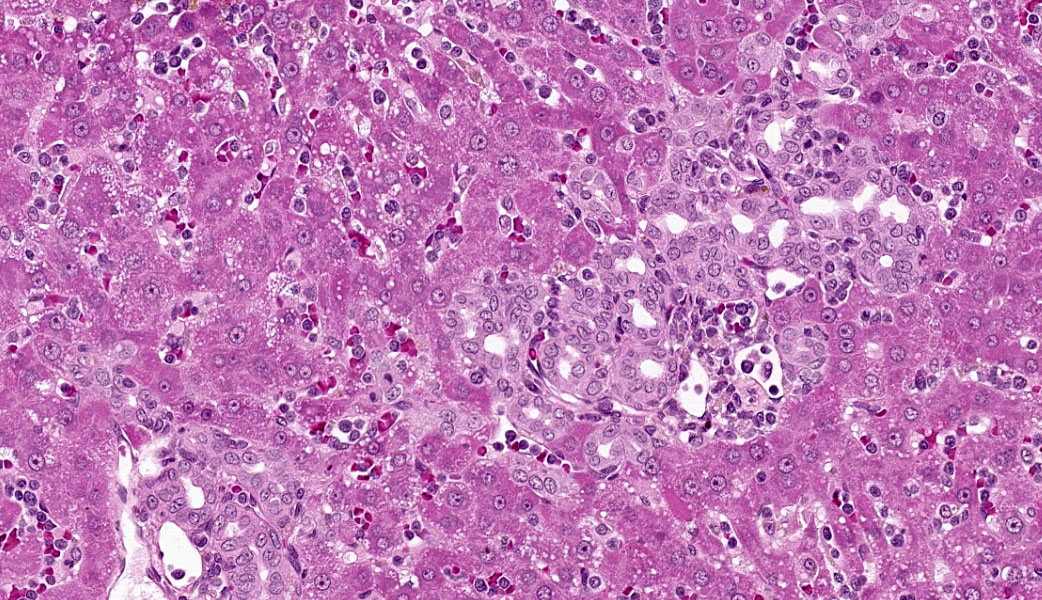
Liver: Diffusely filling portal veins and expanding sinusoids is a neoplastic population composed of round cells with distinct cell borders, small amount of eosinophilic, granular cytoplasm, round to occasional reniform nucleus, clumped chromatin and indistinct nucleolus. Mitotic rate is <1 per hpf.
Diffusely centrilobular hepatocytes are lost, degenerating or necrotic. Diffusely there is lobular atrophy and sinusoids are expanded by previously described neoplastic cells admixed with eosinophilic cellular debris. Multifocally centrilobular kupffer cells and/or hepatocytes contain hemosiderin pigment. Multifocally within portal regions there is an increase in small bile duct profiles (ductular reaction). Rarely, bile duct epithelial cells are degenerative, necrotic or undergoing regeneration.
Contributor's Morphologic Diagnoses:
Liver: Leukemia, large granular lymphocytic
Contributor's Comment:
Large granular lymphocytic (LGL) leukemia, formally identified as mononuclear cell leukemia (MCL), is the main cause of death among Fischer 344 (F344) rats >20 months of age.4 Reported frequency ranges from 10 - 35% with 50% of early deaths is some 2-year studies being caused by this disease.6 LGL leukemia has been reported in other strains of rats.
Wistar-Furth rats have the next highest occurrence with an incidence rate of 15-22%, while the disease is rarely seen in Sprague-Dawley rats (0.6%).7 Naturally occurring LGL leukemia has not been reported in mice.7 Clinical signs are characterized by weight loss, anemia, jaundice, and depression.1 There is usually a concurrent, immune-mediated hemolytic anemia, with thrombocytopenia and clotting abnormalities suggestive of disseminated intravascular coagulation.1 Splenomegaly is the most common characteristic macroscopic finding in rats afflicted with LGL leukemia.4
Histological findings, in general, include diffuse infiltration of the splenic red pulp sinusoids of variable density, infiltration of the hepatic sinusoids with variable centrilobular hepatocellular degeneration, and necrosis and atrophy of hepatic cords and infiltration of the alveolar septa of the lungs.7 Other tissues affected include lymph nodes, adrenal gland, kidney, and bone marrow.
Contributing Institution:
U.S. Army Medical Research Institute of Chemical Defense (https://usamricd.health.mil/)
JPC Diagnoses:
Liver: Large granular cell leukemia.
JPC Comment:
Conference 19 was moderated by the esteemed Dr. Kimberly Whitten, a veteran Department of Defense laboratory animal toxicological pathologist who works for USAMRICD. This first case was a classic entity in F344 rats and one that must be at the forefront of any pathologist's mind in this strain. LGL leukemia in F344 rats closely resembles the NK-cell leukemia seen in humans and, as such, they are used as a research model for this aggressive disease.7 Morphologically, the LGL leukemia cells resemble normal rat LGLs, but there are notable differences in cytotoxicity & surface antigens. Normal rat LGLs usually express surface markers such as CD8, CD2, CD56, and ASGM1, while leukemic LGLs tend to show abnormal or low-level expression of T-cell receptor (TCR) beta chains, may or may not express ASGM1, and can display heterogeneity in markers like CD56.7,9 This suggests that these leukemias may be of a heterogeneous lymphocytic origin, although NK-cell markers predominate.7,9
Natural killer cells are vital, fast-acting lymphocytes of the innate immune system that, unlike most other lymphocytes, identify and destroy virally infected or cancerous cells without prior sensitization. Acting as a first line of defense, they utilize surface receptors to detect absent MHC-I molecules or "stressed" signals on target cells, prompting the NK cells to release cytotoxic granules containing pore-forming enzymes, such as granzyme B.2
Dr. Whitten quizzed participants on other common tumors in F344 rats, which included: testicular interstitial cell tumors, which occur in almost 100% of aged male Fischer rats; mesotheliomas of the tunica vaginalis of the testes, which occur in up to 5% of males and may spread to the peritoneal and pleural cavities; pituitary adenomas, which are seen in 30% of rats and may be prolactin producing, which can result in mammary hyperplasia; mammary gland tumors (primarily mammary fibroadenoma), which occur in roughly 40% of female and 20% of male rats; pheochromocytomas, seen in 30% of male rats in a recent study; and thyroid C-cell carcinomas, occurring in up to 10% of aged rats.3
References:
- Barthold SW, Griffey SM, Percy DH. Rat. In: Pathology of laboratory rodents and rabbits. 4th ed. Ames, IA: John Wiley & Sons, Inc: 2016;166-167.
- Mace EM. Human natural killer cells: Form, function, and development. J Allergy Clin Immunol. 2023;151(2):371-385.
- Sass B, Rabstein LS, Madison R, Nims RM, Peters RL, Kelloff GJ. Incidence of spontaneous neoplasms in F344 rats throughout the natural life-span. J Natl Cancer Inst. 1975;54(6):1449-1456.
- Shiga A, Narama I. Hepatic lesions caused by large granular lymphocytic leukemia in Fischer 344 rats: similar morphologic features and morphogenesis to those of nodular regenerative hyperplasia (NRH) in human liver. Toxicol Pathol. 2015;43:852-864.
- Stefanski SA, Elwell MR, Stromberg PC. Spleen, lymph nodes, and thymus. In: Boorman GA, Eustis LS, Elwell MR, Montgomery CA, MacKenzie WF, ed. Pathology of the Fischer rat: reference and atlas. San Diego, CA: Academic Press;1990:374-379.
- Stromberg PC, Vogtsberger LM. Pathology of the mononuclear cell leukemia of Fischer rats. Vet Pathol. 1983;20:698-708.
- Thomas J, Haseman JK, Goodman JI, et al. A review of large granular lymphocytic leukemia in Fischer 344 rats as an initial step toward evaluating the implication of the endpoint to human cancer risk assessment. Toxicol Sci. 2007;99:3-19.
- Ward JM, Reynolds CW. Large granular lymphocyte leukemia: a heterogeneous lymphocytic leukemia in F344 rats. Am J Pathol. 1983;111:1-10.
- Watters RJ, Liu X, Loughran TP Jr. T-cell and natural killer-cell large granular lymphocyte leukemia neoplasias. Leuk Lymphoma. 2011;52(12):2217-2225.