WSC 22-23
Conf 6
CASE IV:
Signalment:
4 year old, female, African pygmy hedgehog (Atelerix albiventris)
History:
A progressive mass was noted on the right side of the mouth, resulting in halitosis, tooth mobility, exopthalmus, scale loss, anorexia and weight loss. This was refractory to antibiotics (enrofloxacin) and anti-inflammatories (Meloxicam).
Gross Pathology:
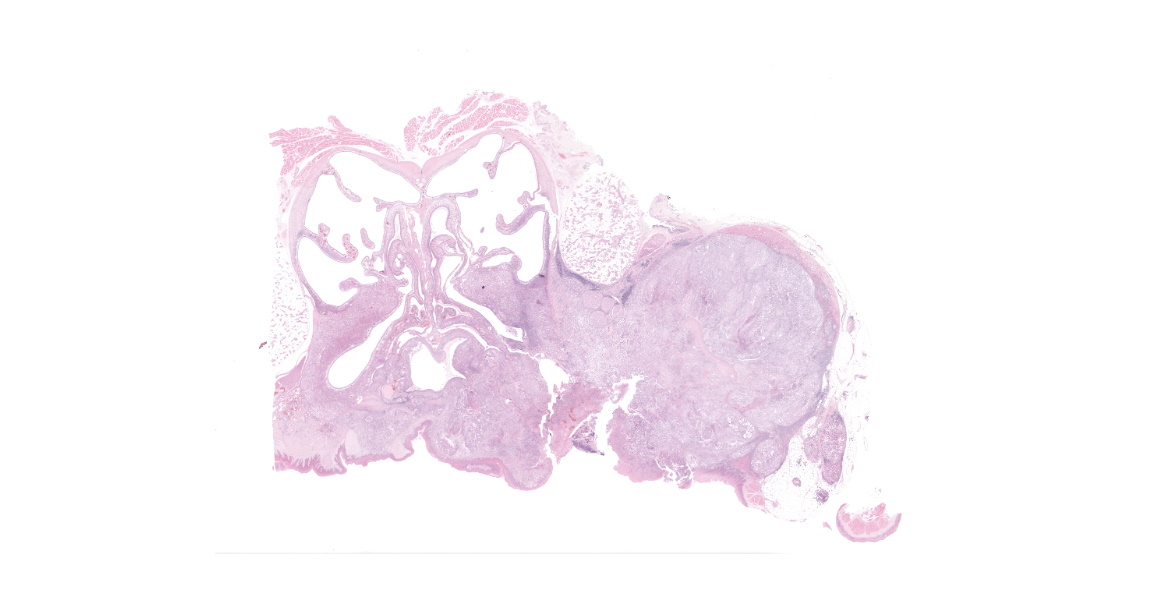
Around the maxillary dental arcade was a poorly demarcated, firm, multilobulated, off-white to tan mass; within the oral cavity, this was approximately up to 25 mm x 14 mm x 12 mm. The surface was ulcerated and only one molar tooth remained, which was very loose. There was a focal area of ulceration of the hard palate (up to 5 mm x 4 mm). The left maxillary dental arcade was missing several cheek teeth and the remaining teeth were loose. On transverse sectioning of the skull, the mass extended dorsally, replacing much of the right side musculature and the maxillary bone, and extending medially, invading the right nasal cavity (the maximum size of the mass was ~25 mm x 14 mm x 20 mm).
Laboratory Results:
No reported laboratory results.
Microscopic Description:
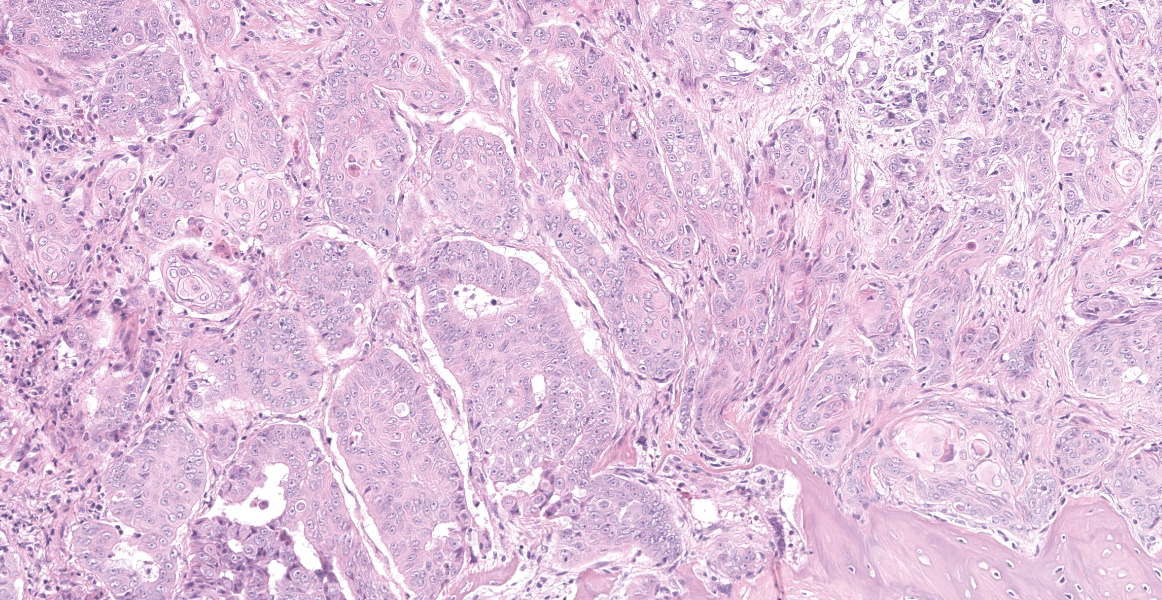
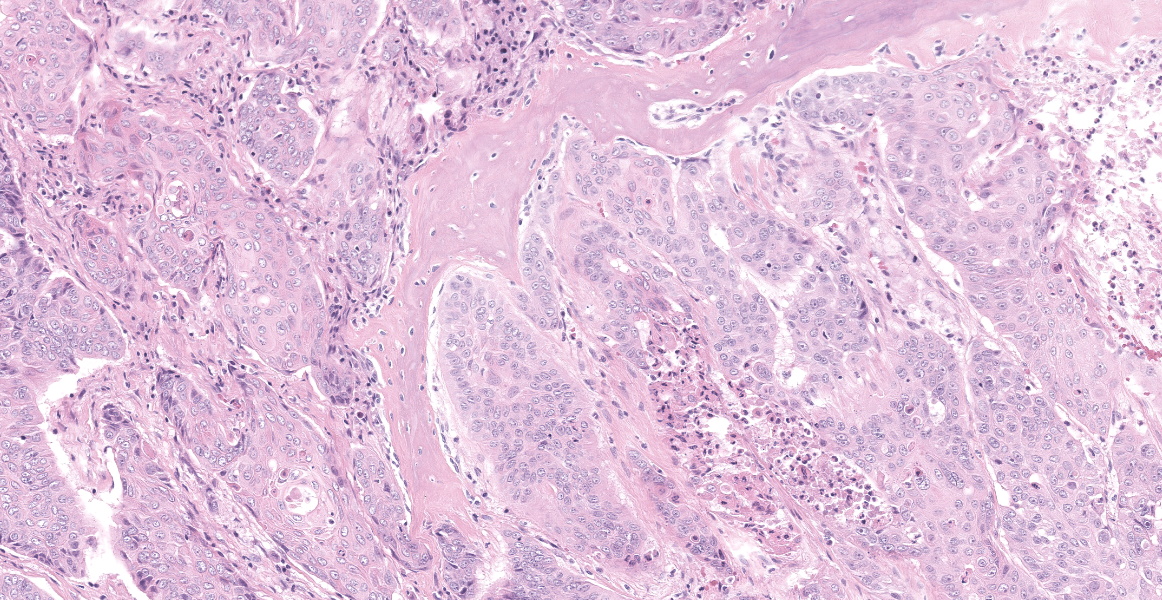
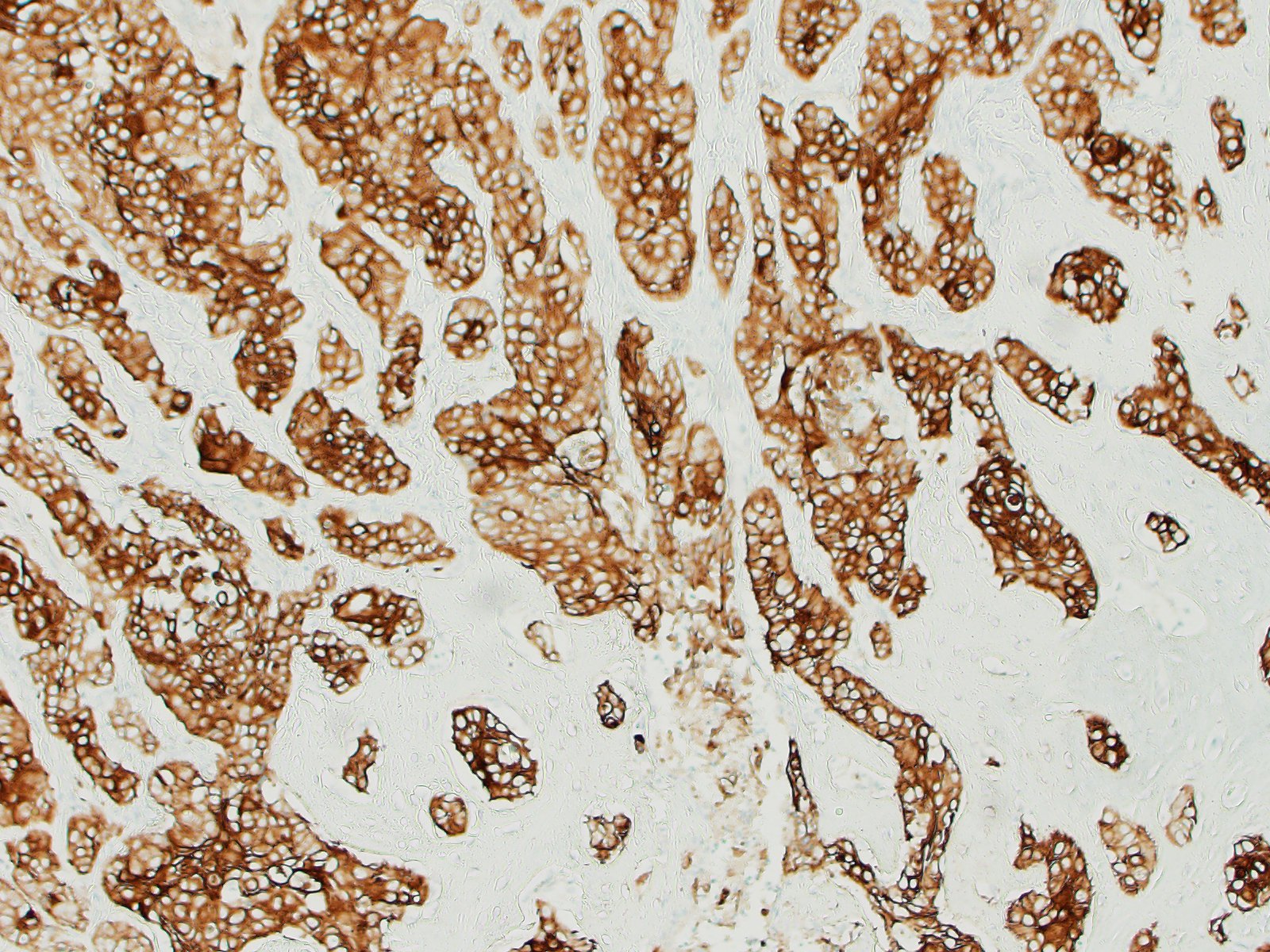
The tissue is markedly effaced and replaced by a poorly demarcated proliferation of atypical squamous epithelial cells arranged in variably sized cords, trabeculae and nests. Individual cells are oval to polygonal, with variably distinct cell borders and a moderate amount of eosinophilic cytoplasm. They have oval to polygonal, hypochromatic, vesicular nuclei, with one or multiple prominent nucleoli. There is moderate anisocytosis and marked anisokaryosis, and frequent binucleate and multinucleate cells. Atypical cells often palisade around the margin of lobules, and central cells often undergo abrupt keratinisation (hypereosinophilic cells with poorly discernible nuclei) or are markedly swollen. There are occasional individualised necrotic cells (hypereosinophilic cells with karyorrhectic or pyknotic nuclei), clusters of necrotic cells with infiltrates of neutrophils, or apoptotic cells (shrunken cells with pyknotic nuclei). The mucosal surface is thinned (eroded), lost (ulcerated) or is degenerate, with marked vacuolated keratinocytes (ballooning degeneration). The subepithelial tissue contains moderate number of lymphocytes, plasma cells and fewer granulocytes.
Contributor’s Morphologic Diagnoses:
Oral squamous cell carcinoma.
Contributor’s Comment:
Oral squamous cell carcinomas (SCCs) are commonly diagnosed in the African pygmy hedgehog, and are the third most common neoplasm in this species, presenting with clinical signs including tooth mobility, edentulism and gingivitis.11 Oral SCCs in African pygmy hedgehogs have been previously described as commonly locally infiltrative with rare metastases, though metastases to distant sites have been reported.5 In addition to oral SCCs, cutaneous SCCs have also been described in African pygmy hedgehogs.2 There are few studies characterizing oral SCCs in African pygmy hedgehogs.
Oral squamous cell carcinomas have been characterized in greater detail in other domestic animals, and is the most common oral tumor in cats and the second most prevalent in dogs.14 These present frequently on the tongue and gingiva, with local invasion, bone invasion and a variable survival, with the greatest mean survival time (MST) following removal of invaded bone.7 Canine SCCs have been classified further into Conventional (82.1% of cases), Papillary and Basaloid (5.95%), Adenosquamous (3.6%) and spindle cell SCCs (2.4%).8 However, there is little data to determine variance in prognosis for these different histological subtypes.7 In any case, spread of oral SCCs to local lymph nodes often occurs late in the disease process4 and local reoccurrence is the most common form of treatment failure.12
Currently, the pathogenesis of oral SCCs is thought to be following long term epithelial hyperplasia from chronic gingivitis irritation.14 In dogs though there may be a tentative link with canine oral papillomavirus (CPV-1) producing papillomas which undergo malignant transformation.13 In humans, multiple factors have been implicated in the development of oral SCCs including environmental factors such as tobacco consumption, alcohol consumption and chronic inflammation which leads to dysplasia of the local area and multiple alterations to tumor suppressor genes (p16, p14ARF, FHIT, RSSFIA and p53) leading to “field cancerization” and development of SCCs.1 “Field cancerization” refers to a theory based on the frequent observation of epithelial dysplasia at the periphery of human invasive oral cancers in situations of chronic exposure to carcinogenic insult, which may leads to increased risk of malignant transformation in the entire area affected by the insult.
Contributing Institution:
Easter Bush Pathology - Royal (Dick) School of Veterinary Studies. https://www.ed.ac.uk/vet/services/easter-bush-pathology
JPC Diagnosis:
Cross section of head: Squamous cell carcinoma.
JPC Comment:
With the growing popularity of hedgehogs as pets, numerous retrospective studies have described the prevalence of neoplasia within captive populations, each with slightly different results. In a study of pet hedgehogs in the US, the most common neoplasms were mammary neoplasia, lymphoma, and oral squamous cell carcinoma.11 In a study in Japan, the most common neoplasms were endometrial stromal tumor, fibrosarcoma, and mammary neoplasms.9 In a study of 63 hedgehogs from the Tai Pei Zoo, the three most common neoplasms were oral squamous cell carcinoma, lymphoma, and pulmonary adenocarcinoma.10 The authors speculated that geographic variability may be due to in-breeding of closed populations due to movement restrictions imposed due to the species’ susceptibility to foot and mouth disease.10
A study of captive hedgehogs in Chile focused specifically on oral masses found that 17 of 27 oral masses were squamous cell carcinoma. These neoplasms were generally ulcerated (13 cases) and occurred most commonly (12 cases) in the caudal aspect of the right maxilla.3 Gingival hyperplasia was the second most common diagnosis (8 cases). Gingival hyperplasia was found in varied location, manifested as pedunculated or sessile masses, and was non-ulcerated.3
Hedgehogs are one of a few non-cloven hooved species which can be naturally infected by foot and mouth disease virus. During a series of in England in the 1940s, several infected wild hedgehogs were found with vesicles on the feet, and researchers subsequently demonstrated that cattle could experimentally infect hedgehogs.6 The authors concluded that hedgehogs had some role in spreading infection between cattle. 6
References:
- Choi, S. & Myers, J. N. Molecular pathogenesis of oral squamous cell carcinoma: Implications for therapy. Dent. Res. 87, 14-32 (2008).
- Couture, É. L., Langlois, I., Santamaria-Bouvier, A. & Benoit-Biancamano, M. O. Cutaneous squamous cell carcinoma in an African pygmy hedgehog (Atelerix albiventris). Vet. J. 56, 1275-1278 (2015).
- Del Aguila G, Torres C, Carvallo FR, Gonzolaz CM, Cifuentes FF. Oral masses in African pygmy hedgehogs. Jour Vet Diag Invest. 2019; 31(6): 864-867.
- Grimes, J. A. et al. Histologic evaluation of mandibular and medial retropharyngeal lymph nodes during staging of oral malignant melanoma and squamous cell carcinoma in dogs. Am. Vet. Med. Assoc. 254, 938-943 (2019).
- Heatley, J., Mualdin, G. & Cho, D. A Review of Neoplasia in the Captive African Hedgehog (). Avian Exot. Pet Med. 14, 182-192 (2005).
- McLaughlan JD, Henderson WM. The occurrence of foot-and-mouth disease in the hedgehog under natural conditions.J Hyg (Lond). 1947; 45(4): 474-479.
- Meuten D.J. Tumors in domestic animals. 5th Ames, IO: John Wiley & Sons, Inc. 2002.
- Nemec, A., Murphy, B., Kass, P. H. & Verstraete, F. J. M. Histological Subtypes of Oral Non-tonsillar Squamous Cell Carcinoma in Dogs. Comp. Pathol. 147, 111-120 (2012).
- Okada K, Kondo H, Sumi A, Kagawa Y. A retrospective study of disease incidence in African pygmy hedgehogs (Atelerix albiventris).
- Pei-Chi H, Jane-Fang Y, Lih-CHiann W. A Retrospective Study of the Medical status on 63 African Hedgehogs (Atelerix albiventris) at the Taipei Zoo from 2003 to 2011. J Exot Pet Med. 2015; 24:105-111.
- Raymond, J. T. & Garner, M. M. Spontaneous tumours in captive African hedgehogs (Atelerix albiventris): A retrospective study. Comp. Pathol. 124, 128-133 (2001).
- Riggs, J. et al. Outcomes following surgical excision or surgical excision combined with adjunctive, hypofractionated radiotherapy in dogs with oral squamous cell carcinoma or fibrosarcoma. Am. Vet. Med. Assoc. 253, 73-83 (2018).
- Thaiwong, T. et al. Malignant transformation of canine oral papillomavirus (CPV1)-associated papillomas in dogs: An emerging concern? Papillomavirus Res. 6, 83-89 (2018).
- Uzal F, Plattmer B. Alimentary System. In: Maxie MG, ed. Jubb, Kennedy and Palmer’s Pathology of Domestic Animals. Vol 2. 6th Philadelphia, PA: Elsevier Saunders. 2016: 25-26.