Conference 17, Case 1
Signalment:
Female Indian ringneck parrot/parakeet (Psittacula krameria)
History:
Trouble breathing, wings out.
Gross Pathology:
There was moderate gelatinous hydrothorax and ascites. The lungs were congested and edematous.
Laboratory Results:
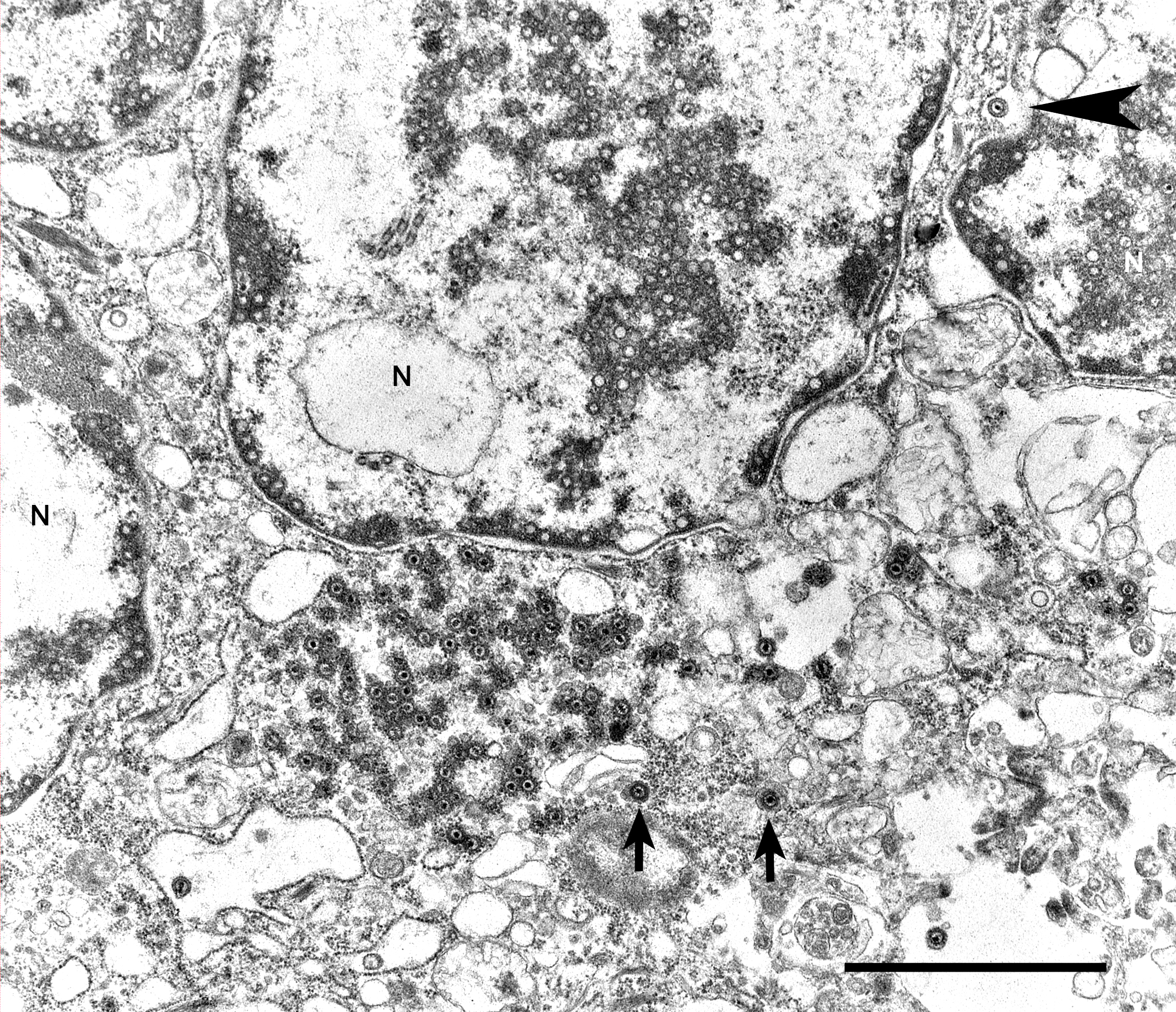
Transmission electron microscopy - lung: Intranuclear and intracytoplasmic herpesviral replication complexes
- Psittacine herpesvirus 1 PCR - lung: negative
- Herpesvirus PCR and sequencing: positive - Psittacid herpesvirus 5
Microscopic Description:
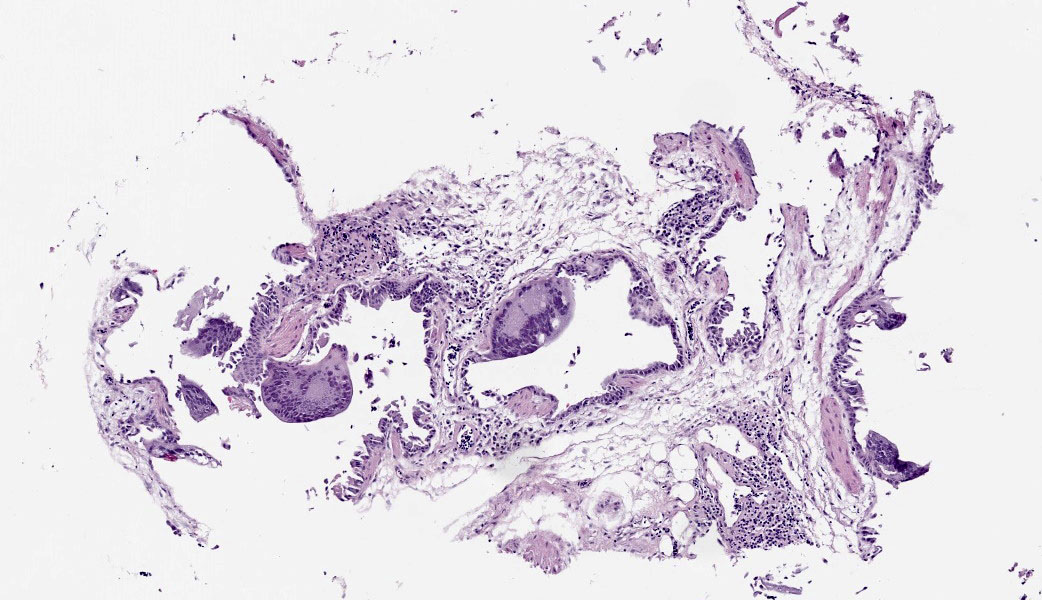
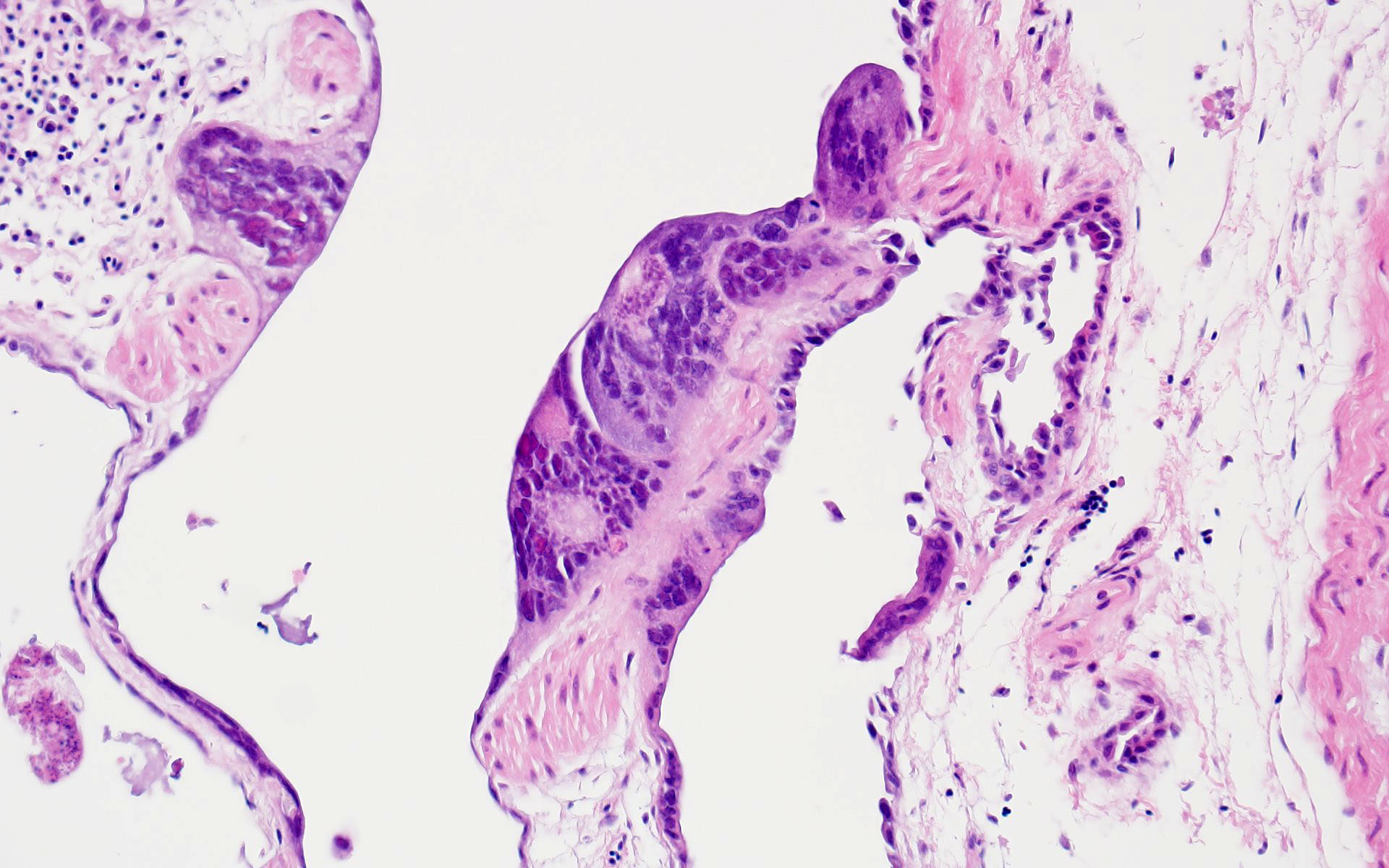
The bronchi and parabronchi contain numerous large syncytia with large amphophilic-to-eosinophilic intranuclear inclusions that marginalize the chromatin. Bronchi and parabronchi also often contain foamy macrophages, pale basophilic mucus, homogeneously eosinophilic edema fluid, fibrin, cellular debris, and sloughed necrotic epithelial cells.
Contributor’s Morphologic Diagnosis:
Lung: Pneumonia, histiocytic, with syncytia and intranuclear inclusion bodies
Contributor’s Comment:
Pulmonary lesions in this parrot were consistent with a herpetic pneumonia (cause: Psit-tacid alphaherpesvirus 5). Herpesviridae is a large family of double-stranded DNA viruses that can infect a wide variety of animal species, including fish, mollusks, reptiles, birds and mammals.1,3 A hallmark of this family of viruses is persistent infection (or latency) with intermittent or continuous shedding. Herpesviruses can be sub-classified into alpha-, beta-, or gamma-herpesviruses. The subfamily Al-phaherpesvirinae is further subdivided into the following genera: Iltovirus, Scutavirus, Simplexvirus, Varicellovirus, and Mardivirus.
The subfamily Betaherpesvirinae is further subdivided into the following genera: Cytomegalovirus, Muromegalovirus, Proboscivirus, Quwivirus, and Roseolovirus. Similarly, the subfamily, Gammaherpesvirinae is also subdivided into several genera – Lymphocryptovirus, Bossavirus, Macavirus, Manticavirus, Patagivirus, Percavirus, and Rhadinovirus.1
Herpesviruses of veterinary significance and their classification:3
- Subfamily Alphaherpesvirinae: Bovine herpesvirus 1 (Infectious bovine rhinotra-cheitis virus, Infectious pustular vulvo-vaginitis virus), Bovine herpesvirus 2 (Mammillitis, Pseudo-lumpy skin disease virus), Bovine herpesvirus 5 (Bovine en-cephalitis virus), Canid herpesvirus 1, Caprine herpesvirus 1, Cercopithecine herpesvirus 1 (B virus disease of ma-caques), Cercopithecine herpesvirus 9 (Simian varicella virus), Equid herpesvi-rus 1, Equid herpesvirus 3 (Equine coital exanthema virus), Equid herpesvirus 4 (Equine rhinopneumonitis virus), Felid herpesvirus 1 (Feline viral rhinotracheitis virus), Gallid herpesvirus 1 (Avian infectious laryngotracheitis virus), Gallid her-pesvirus 2 (Marek’s disease virus), Human herpesvirus 1, Human herpesvirus 2, Hu-man herpesvirus 3 (Varicella-zoster vi-rus), Saimiriine herpesvirus 1 (Herpesvi-rus tamarinus), Chelonid herpesvirus 5
- Subfamily Betaherpesviriane: Murid herpesvirus 1 and 2, Elephantid herpesvirus (Endotheliotropic elephant herpesvirus), Suid herpesvirus (Porcine cytomegalovirus), Macacine herpesvirus 3 (Rhesus cytomegalovirus)
- Subfamily Gammaherpesvirinae: Malignant catarrhal fever (cause: Alcelaphine herpesvirus 1, Ovine herpesvirus 2), Bovine herpesvirus 4, Equid herpesvirus 2, Equid herpesvirus 5, Equid herpesvirus 7 (Asinine herpesvirus 2), Human herpesvi-rus 4 (Epstein-Barr virus), Saimiriine her-pesvirus 2 (Herpesvirus saimiri), Otarine herpesvirus 1
There are a number of herpesviruses of signif-icance in birds including Gallid alphaherpes-virus 1 (GaAHV1; causative agent of infectious laryngotracheitis), Gallid alphaherpesvi-rus 2 (GaAHV2; causative agent of Marek’s disease), Anatid alphaherpesvirus 1 (AnAHV1; causative agent of duck viral en-teritis), Psittacid alphaherpesvirus 1 (PsAHV1; causative agent of Pacheco’s dis-ease and papillomatosis), and Columbid al-phaherpesvirus 1 (CoAHV1; pigeon herpesvi-rus; fatal disease in hawks, owls, and falcons). In psittacines, at least 5 herpesviruses have been named; all of the aforementioned viruses cluster in the genus Iltovirus.2
- Psittacid alphaherpesvirus 1 (PsAHV1)
- Provisionally named Psittacid alphaherpesvirus 2 (PsAHV2)
- Provisionally named Cacatuid alphaherpesvirus 1
- Provisionally named Cacatuid alphaherpesvirus 2
Provisionally named Psittacid alphaherpesvirus 3 (PsAHV3)
Provisionally named Psittacid alphaherpesvirus 5 (PsAHV5)
Respiratory herpesviruses have been de-scribed in Indian ringneck parrots as early as the 1990s.4,8 Similar gross and microscopic le-sions have been reported in psittacines infected with PsAHV3 and PsAHV5, including the formation of large syncytia and intranu-clear inclusions throughout the respiratory tract.2,6,7 Thus far, PsAHV5 has been detected in Indian ringneck parakeets, Alexandrine parakeets, Bourke’s parrots, and possibly eclectus parrots. Concurrent infections, such as Aspergillosis, are common.2
Contributing Institution:
California Animal Health and Food Safety Laboratory (San Bernardino Branch) https://cahfs.vetmed.ucdavis.edu/
JPC Diagnoses:
- Tubular reproductive tract, air sac, lung: Epithelial necrosis, acute, multifocal, moderate, with intranuclear viral inclusions and numerous syncytia.
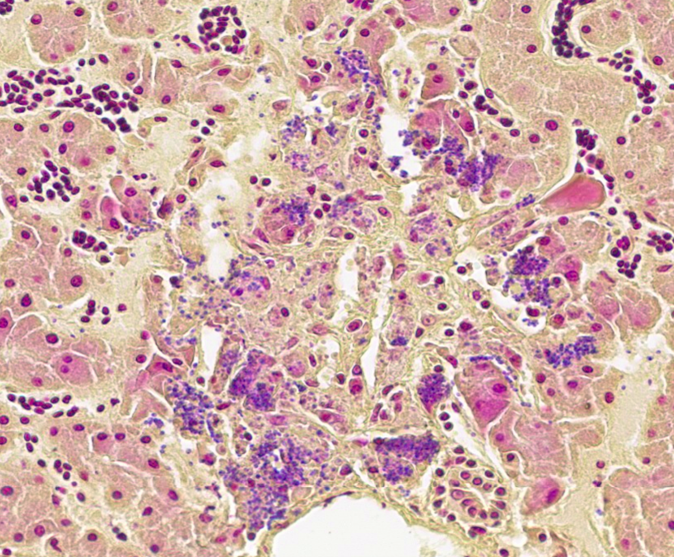
- Liver: Hepatitis, necrotizing, acute, multifocal, random, moderate with microbes consistent with microsporidia.
JPC Comment:
The JPC’s own Dr. Elise LaDouceur moder-ated this year’s avian-focused seventeenth conference. This first case provided participants with a panoply/salmagundi/”dog’s breakfast” of tissues to sort through to achieve a diagnosis. The characteristic intranuclear viral inclusion bodies of herpesvirus were best seen in the air sac respiratory epithelium, which also rewarded conference-goers with some exceptional viral syncytia. Of the potential herpesviral culprits, psittacine al-phaherpesviruses (PsAHV) 1, 3, and 5 have been reported in psittacine species, including Indian ringneck parakeets, and PsAHV-5 was isolated by the contributor in this case. The contributor provided some beautiful electron microscopy (EM) photos from this bird that demonstrated intranuclear herpesviral virions, with some virions budding from the nuclear envelope to become enveloped themselves, and others hanging around the rough endo-plasmic reticulum (RER) to acquire additional proteins. The JPC is grateful to the contributor for providing these excellent educational images!
In addition to the respiratory epithelium, a tubular structure adjacent to the ovary was also affected by PsAHV-5 (i.e., had syncytia and epithelial necrosis). The smooth muscle wall and cellularity of this structure is consistent with either oviduct or remnant Wolffian duct. This suggests that PsAHV-5 may have a broader epitheliotropism beyond respiratory epithelium.
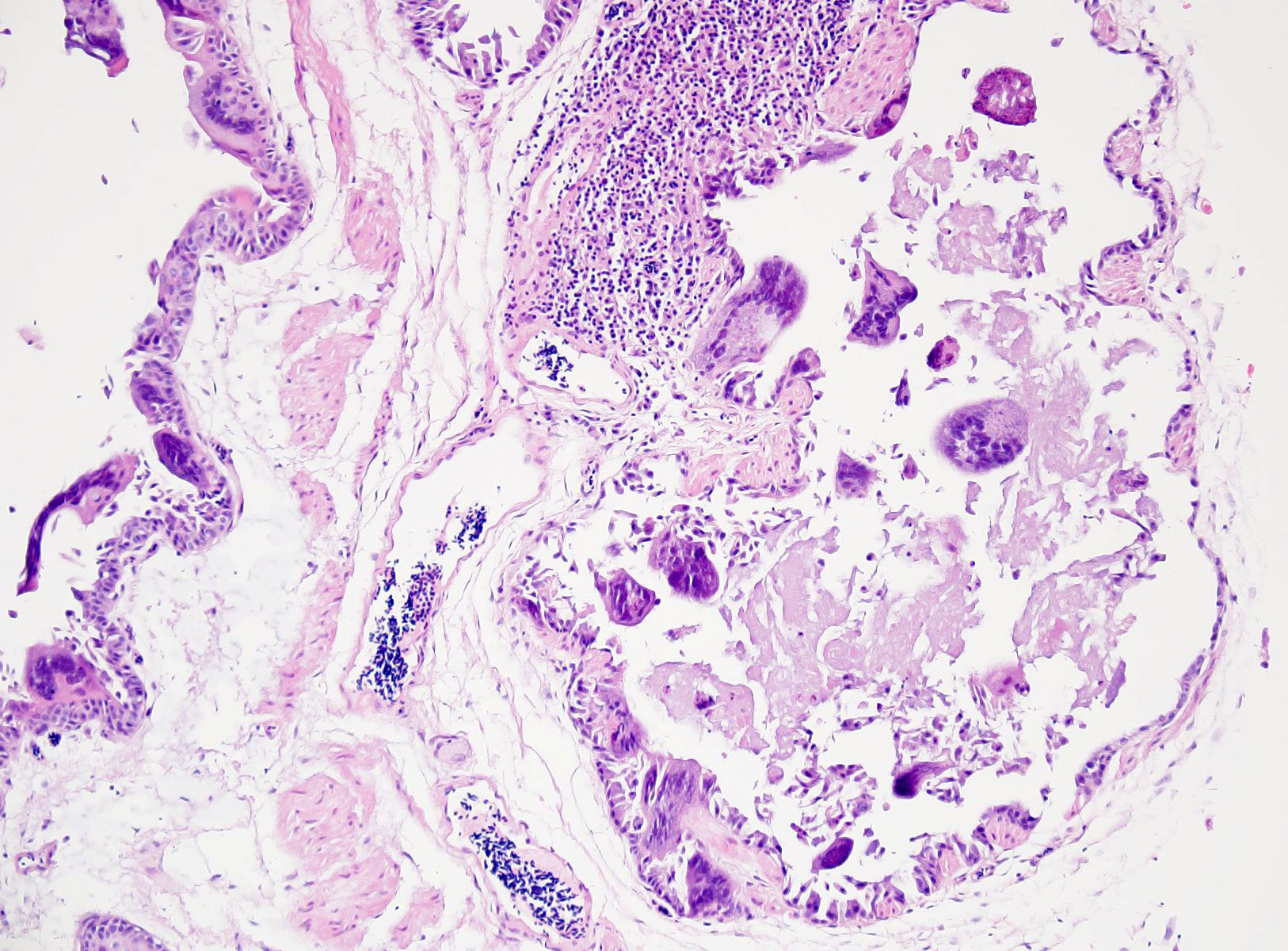
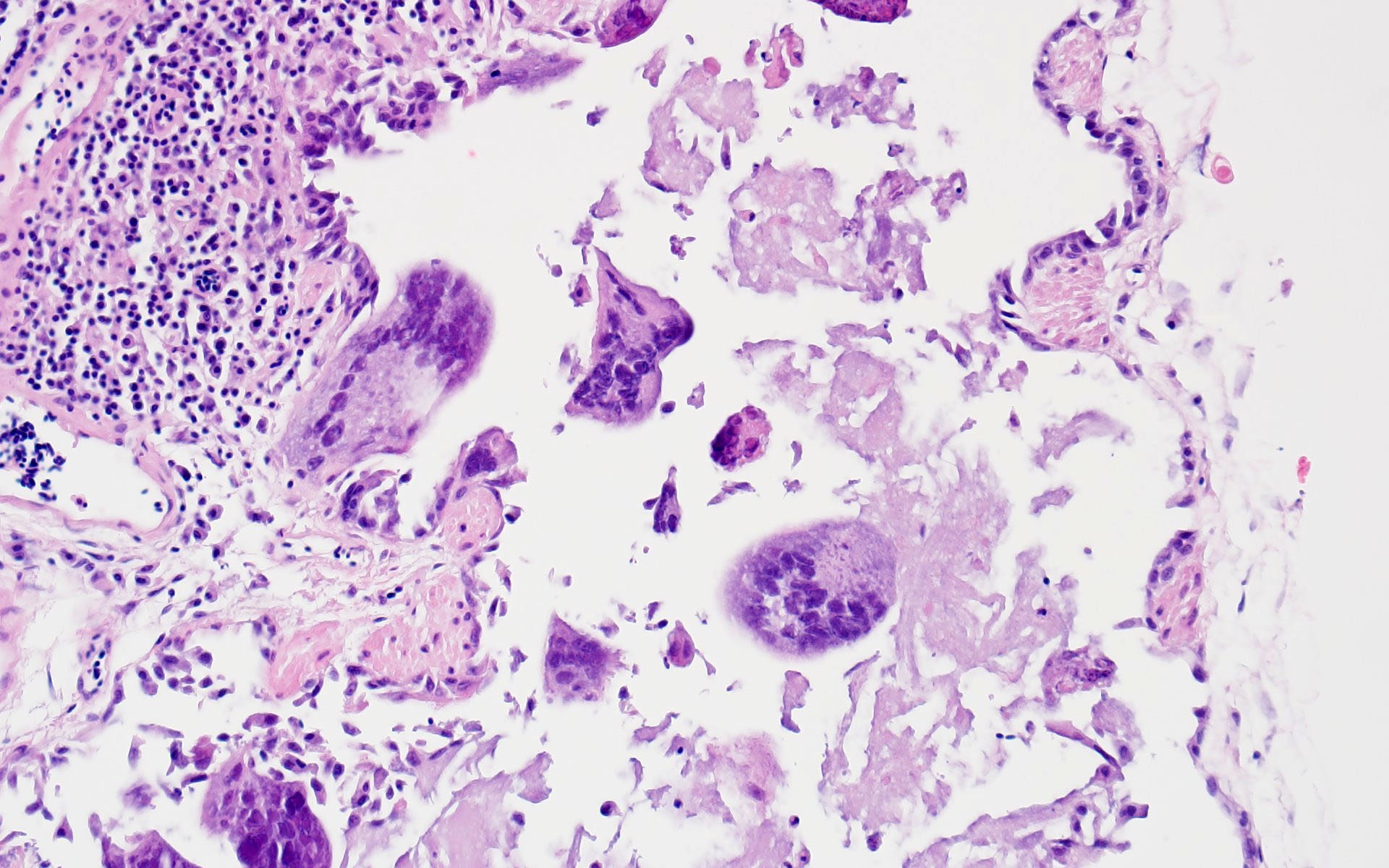
The liver had multifocal, random hepatocellular necrosis centered on microbes that were intracellular (within hepatocytes), round to ovoid, and 1.5-2.5µm with an eccentric clearing. These organisms were gram-positive, argyrophilic on GMS, and positively staining on Giemsa; PAS and acid-fast stains were negative. Conference participants discussed at length the potential identity of these organisms. The primary differential is Encephalitozoon hellum, which is a microsporidian parasite in psittacines. E. hellum is an intracellular parasite that is ovoid, 1.5-2.5 µm, and has a predilection for the liver.5 It is most common in immunosuppressed animals, which was likely in this case given the herpesvirus infection. Other differentials raised by participants included chlamydiosis, which can cause intra-cellular bacterial inclusions in the liver, but the individualized organisms were too large for chlamydiosis. Additionally, Chlamydia spp. would be expected to be gram-negative (unlike the gram-positive organisms in this case).5 Candidiasis was also considered; how-ever, yeasts should be positive for PAS (unlike the PAS negative organisms in this case) and is typically larger than 1.5µm.5 Ultimately, confirmatory diagnosis of E. hellum would require molecular investigation or ultrastructural analysis (transmission electron microscopy) revealing polar filaments.5
Participants also noted the characteristic “freeze-thaw” artifact of the liver that is best appreciated at low magnification and causes acicular clefts filled with pale eosinophilic fluid. This is a classic artifact to be aware of!
References:
- Gatherer D, Depledge DP, Harley CA, et al. ICTV virus taxonomy profile: Herpes-viridae J Gen Virol 2021;102:001673.
- Henderson EE, Streitenberger N, Asin J, et al. Psittacid alphaherpesvirus 5 infection in Indian ringneck parakeets in southern California. J Vet Diagn Invest. 2023;35(1):67-71.
- Knowles DP. Herpesvirales. In: MacLachlan NJ, Dubovi EJ ed. Fenner’s Veterinary Virology. 4th Elsevier 2011:179-202.
- Lazic T, Ackermann MR, Drahos JM, Stasko J, Haynes JS. Respiratory herpes-virus infection in two Indian Ringneck parakeets. J Vet Diagn Invest. 2008;20:235–238.
- Schmidt RE, Struthers JD, Phalen DN. Liver. In: Pathology of Pet and Aviary Birds. 3rd ed. John Wiley & Sons; 2024: 197-238.
- Shivaprasad HL, Phalen A novel her-pesvirus associated with respiratory dis-ease in Bourke’s parrots (Neopsephotus bourkii). Avian Pathol. 2012;41:531–539.
- Sutherland M, Sarker S, Raidal SR. Mo-lecular and microscopic characterisation of a novel pathogenic herpesvirus from In-dian ringneck parrots (Psittacula kra-meri). Vet Microbiol. 2019;239:108428.
- Tsai SS, Park JH, Hirai K, Itakura Her-pesvirus infections in psittacine birds in Japan. Avian Pathol.1993;22:141–156.