CASE 2: 20V-12504-37 (4152969-00)
Signalment:
30-year-old, intact female, 72.4kg Harbor Seal (Phoca vitulina)
History:
This seal had an on/off history of head tremors for more than 6 months starting on May 2019. These head-tremors/bobbling apparently resolved in November 2019; the head twitching was partially attributed to stress and presumptive West Nile virus (WNV) infection based on remarkably elevated titers (>6000). In March 2020 the abnormal head movement returned, on physical examination increased respiratory effort was noted; animal started on antibiotics, antifungals and anxiolytics (diazepam); this animal has had a chronic (since 2010) dental disease with previous extractions at former institution. The affected seal was Immobilized on 03/03/2020 for exam, bronchoscopy, CT scan. Bronchoscopy only revealed moderate to severe congestion of proximal tracheal mucosa. CT scan of head and thorax obtained; Iohexol (+/- 200mls) passed through a jugular catheter for thoracic CT scan but no enhancement noted and there seems likely that there was extravasation on this area, however during necropsy no evidence of SC Fluid was observed. Euthanasia was ultimately electe?d at this time.
Gross Pathology:
There are appropriate subcutaneous and visceral fat stores, there are no signs of dehydration, and the body is in fresh postmortem condition. The cribriform plate is grossly unremarkable. Peripheral lymph nodes, including the axillary, subscapular, and mesenteric lymph nodes, are all prominent. The mesenteric lymph node chain is approximately 1.5 cm in diameter and over 15 cm long. The ventral 50% of the lungs in all fields are atelectatic. There is mild thickening of the hepatic capsule with an irregular margin. There is similar thickening of the renal capsules bilaterally. The gingiva is mildly reddened and inflamed. There is scant, mucoid digesta present within the stomach and small intestines; there are well-formed feces in the descending colon. The brain and spinal cords are unremarkable, grossly.
Laboratory results:
|
Test |
Pathogens |
Result |
|
|
Toxoplasma spp. |
1:160 |
|
Sarcocystis spp. |
<1:40 |
|
|
Neospora spp. |
<1:40 |
|
|
West Nile Virus |
>1:6144 |
|
|
Eastern equine encephalitis virus |
Negative |
|
|
Western equine encephalitis virus |
Negative |
|
|
Venezuelan encephalitis virus |
Negative |
|
|
Canine distemper virus |
>1:512 |
|
|
Phocine distemper virus |
1:128 |
|
|
|
Toxoplasma spp. |
Negative |
|
Sarcocystis spp. |
Negative |
|
|
Neospora spp. |
Negative |
|
|
Morbillivirus |
Negative |
|
|
West Nile virus |
Positive |
|
|
Agar gel immunodiffusion test
|
Leptospira spp. |
1:100 |
|
Blastomyces spp. |
Negative |
Microscopic description:
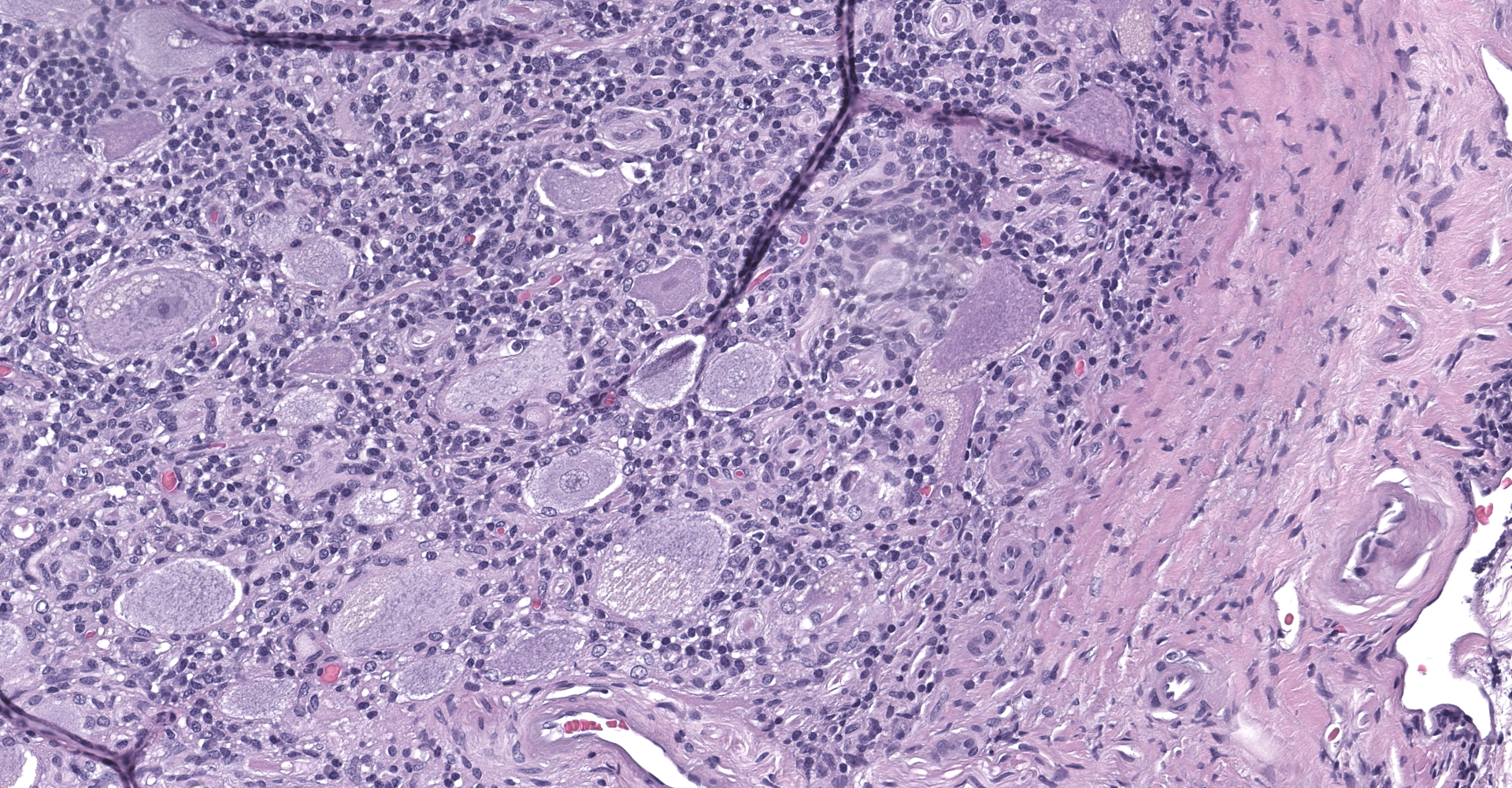
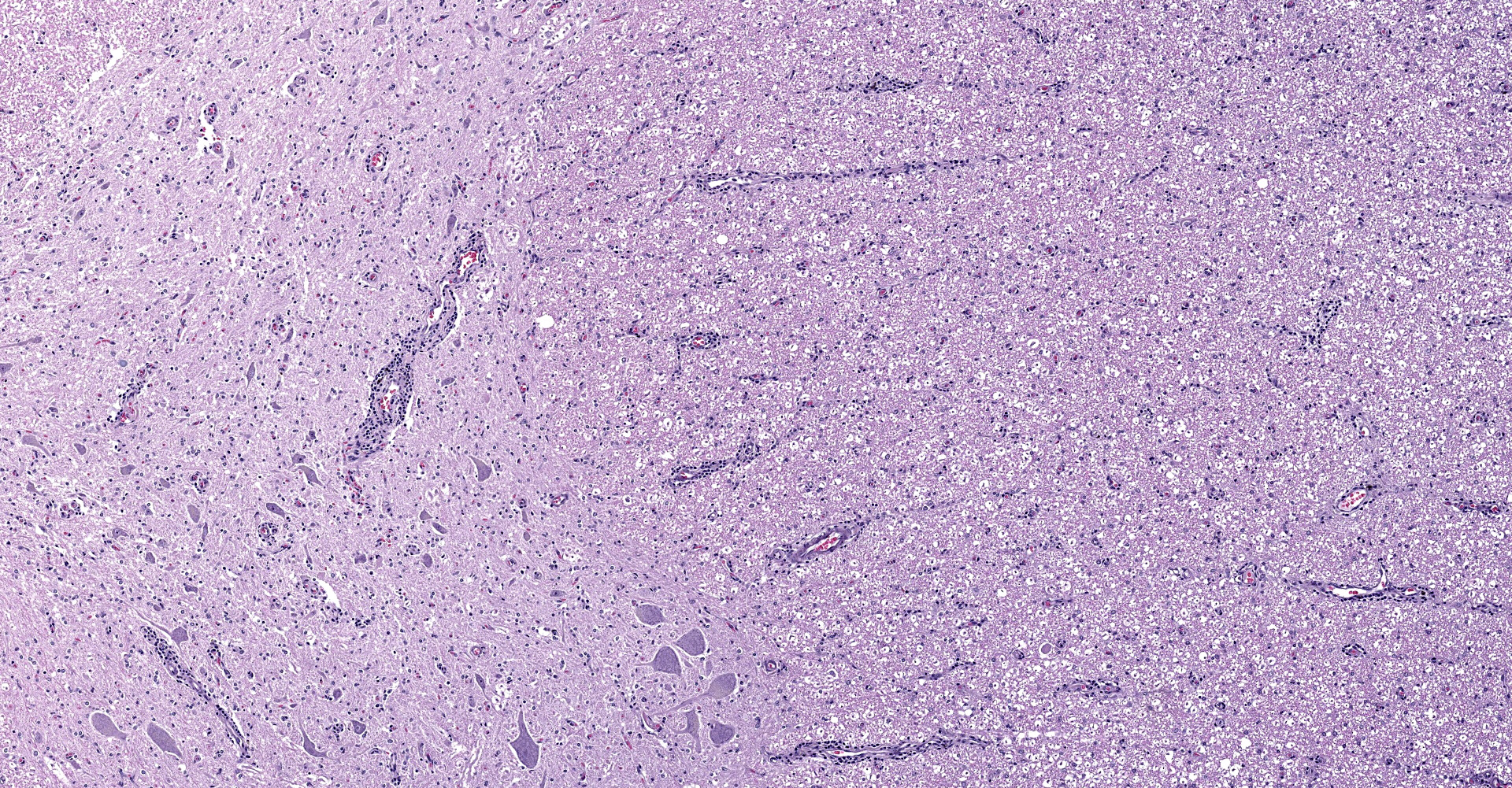
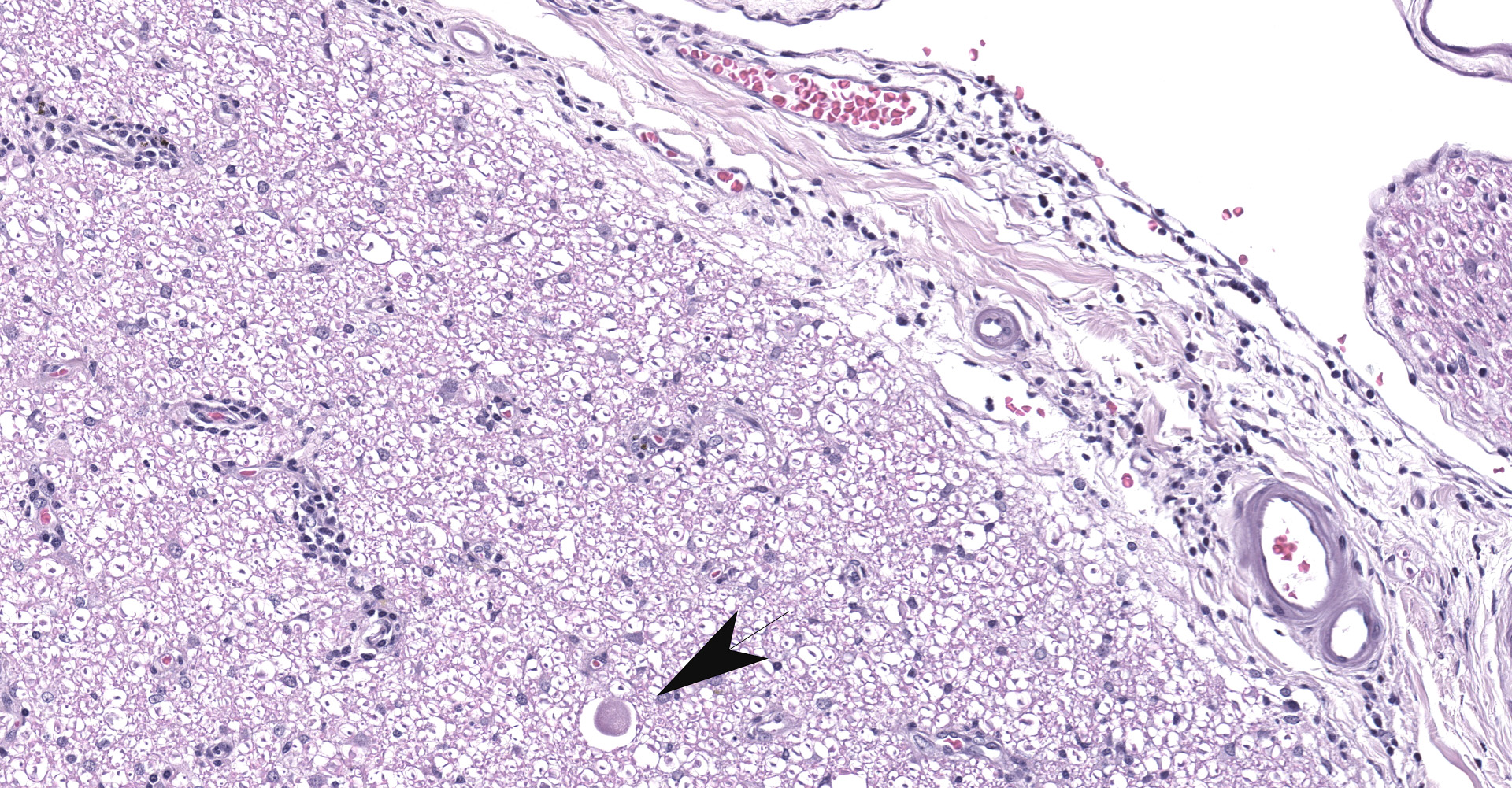
Multifocally in the white and gray matter, there are perivascular infiltrates of moderate numbers of lymphocytes, plasma cells and occasional macrophages. There are multifocal areas of rarefaction in the white matter neuropil creating discrete cavitary spaces in some of which is cellular debris (digestion chamber). There are dilated myelin sheaths with swollen hypereosinophilic axons (spheroids) scattered throughout the neuropil. These are minimal/mild multifocal areas of proliferation of glial cells. The spinal meninges are diffusely expanded by extravasated erythrocytes and frequently contain moderate numbers of scattered interstitial and perivascular infiltrates of lymphocytes, plasma cells and histiocytes. The gray matter contains multifocal glial nodules and frequent neurons contain small to moderate amounts of intracytoplasmic perinuclear yellowish granular material (ceroid-lipofuscin). Within the ganglion, there is locally extensive area of lymphoplasmacytic infiltrates and neuronal ganglion degeneration characterized by shrunken and infrequently dispersed Nissl substances (central chromatolysis). In the same area, adjacent neuronal ganglion is hypercellular with increased number and prominence of satellite cells dissecting perineurium and endoneurium (satellitosis). There are also multifocal areas of mild swollen axons. The neuronal ganglions also contain variable amounts of ceroid-lipofuscin pigments and intracytoplasmic vacuoles that represent incidental aging changes.
Contributor's morphologic diagnosis:
Spinal cord & Ganglia: Moderate to severe, subacute to chronic, multifocal lymphoplasmacytic and histiocytic meningomyelitis and moderate to severe, subacute, locally extensive lymphoplasmacytic ganglioneuritis with satellitosis
Contributor's comment:
West Nile Virus (WNV) is one of the arthropod-borne encephalitides (arboviruses), and is a positive-sense, single-stranded, RNA virus that belongs to genus Flavivirus, family Flaviviridae.9 The virus is distributed worldwide with a wild host range. A variety of Culex sp. mosquitos are the primary vector, but ticks are also capable of harboring the virus. Several bird species are highly susceptible to infection, while domestic poultry and psittacines are generally resistant to fatal infections. A member of Covidae (crow family) and American Robin play a key role in bird-mosquito cycle and act as amplifying hosts.1 A disease transmission through prey or contaminated water have been reported in additional to horizontal transmission.4 WNV is composed of a virus envelope from the host cell membrane with two integral transmembrane glycoprotein E which play a crucial role in a viral pathogenesis. The E2 glycoprotein is involved with attachment to target cells while the E1 glycoprotein has a role in entering host target cells via endocytosis. Within host cells, after inoculation by mosquitoes, the virus propagates in cutaneous dendritic cells, Langerhans cells, fibroblasts, endothelial cells, and keratinocytes, and spreads to regional lymph nodes.1 The virus can cross the blood brain barrier and enter the brain hematogenously, and possibly also through a retrograde axonal transportation.1 Neurons and microglial cells are a vulnerable target for the virus and contribute to encephalitis while mononuclear cells are important targets and contribute to the systemic spread.
In marine mammals, WNV infection has been only reported in a harbor seal (Phoca vitulina)2 and a killer whale (Orcinus orca).8 In this case, the histopathological findings and gross findings are somewhat similar to those previously described in the harbor seal2 and the killer whale,8 which includes non-suppurative encephalomyelitis, gliosis, with occasional neuronal degeneration and necrosis. In addition to previous reports, our case demonstrates intense lymphoplasmacytic ganglioneuritis. In terms of serum neutralization titer, according to Cornell WNV diagnostic laboratory a titer of >6144 is confirmatory of WNV infection and disease based on what is seen in other mammals. Additionally, a serological screening test has been reported in non-fatal WNV infection in a seal at the Detroit zoo with signs of neurologic dysfunction of 4 days' duration2 and bottle nose dolphins (Tursiops truncatus) from the Indian River Lagoon even though WNV-associated disease in these animals has not been reported.7 Other differential diagnoses for encephalomyelitis and/or meningoencephalitis in seals are well summarized in Del Piero et al. (2006)2 including phocine morbillivirus, influenza A8 and B11 orthomyxoviruses, Toxoplasma gondii, and Sarcocystis neurona. Therefore, the combination of routine histopathological examination (multiple sections of brain, brain stem, and spinal cord) and ancillary laboratory tests such as immunohistochemistry, viral isolation, serum neutralization test and qPCR increase the specificity and sensitivity for WNV identification.2 This report also implicates the WNV infection in marine mammals that can carry a potential risk for zoonotic transmission to zookeepers and veterinarians.
Contributing Institution:
Oregon State University Magruder Hall Rm. 134 700 SW 30th Street Corvallis, OR 97331
https://vetmed.oregonstate.edu/diagnostic
Oregon Zoo Veterinary Hospital Portland, OR 97221-2799
https://www.oregonzoo.org/discover/new-zoo/veterinary-medical-center
JPC diagnosis:
1. Ganglion: Ganglioneuritis, lymphoplasmacytic, multifocal, moderate with mild neuronal degeneration and satellite cell hyperplasia.
2. Spinal cord: Meningomyelitis, lymphoplasmacytic, diffuse, mild to moderate.
3. Cerebellum: Leptomeningitis, lymphoplasmacytic, multifocal, mild.
JPC comment:
In the decades since its introduction to the New World in 1999, the geographic distribution of West Nile Virus has expanded tremendously. The accepted virus cycle is between birds and mosquitos, but as noted by the contributor, mammalian species may be infected as well. Beyond the discussion of infection and the development of disease, research questions remain as to which species may serve as competent hosts for the virus. Some species, such as the fox squirrel (Sciurus niger) develop a viremia at titers suggesting it may be a moderately competent host for mosquito infection, but a great many others' capacity for viral propagation are currently unknown.5
Tracking the geographic spread of the virus and being able to quickly identify small variations in the virus provides additional information for public health professionals, researchers, educators, and students. A program that tracks the evolution of West Nile Virus in the Americas is called Nextstrain (nextstrain.org/WNV/NA), which implements a GIS system and phylogenetic analyses with user tools for display variable modifications. This tool is potentially useful for demonstrating the need to dedicate resources in affected areas to prepare for and combat this virus.3
Research in animal models has shown that WNV infection provokes both innate and adaptive immune response in the host. TLR3 and TLR7 (endosomal) and RLR (RIG like receptors) ultimately induce the synthesis of type 1 interferons (IFN) and proinflammatory cytokines. Type III interferons (IFN-g) promote the integrity of the blood brain barrier, limiting entry of WNV to the CNS. g-d T cells play a role in early disease response by secreting IFN-g, promoting dendritic cell maturation, and priming of T lymphocytes. B lymphocytes and a good humoral response helps control the spread of the infection but is not sufficient alone. A combination of complement, a-b T lymphocytes, CD4+ T lymphocytes, and CD8+ lymphocytes are required to eliminate the infection and prevent viral persistence.6
During conference discussion, the moderator indicated that a more specific name for ganglionic satellite cells is the term amphicytes. Additionally, an aggregate of satellite cells with a likely loss of ganglionic neuronal body is a Nageotte body. In order to help differentiate satellite cells from inflammatory cells, immunohistochemistry may be performed, with satellite cells demonstrate positive immunoreactivity to CD204, CD18, IBA1, GFAP, SOX2, nestin, and a few others.
References:
1. Cantile C, Youssef S. Nervous system. In: Maxie MG, ed. Jubb, Kennedy, and Palmers Pathology of Domestic Animals. Vol 1. 6th ed. St. Louis, MO: Elsevier; 2016:374-375.
2. Del Piero F, Stremme DW, Habecker PL, Cantile C: West Nile Flavivirus Polioencephalomyelitis in a harbor seal (Phoca vitulina). Vet Pathol 2006:43(1):58-61.
3. Hadfield J, Brito AF, Swetnam DM, et al. Twenty years of West Nile virus spread and evolution in the Americas visualized by Nextstrain. PLOS Pathogens. 2019;15(10):e1008042.
4. Miller AD, Zachary JF. Nervous System. In: McGavin MD, Zachary JF, eds. Pathologic Basis of Veterinary Disease. 6th ed. St. Louis, MO: Elsevier Mosby; 2017:876-877.
5. Root JJ, Bosco-Lauth AM. West Nile Virus Associations in Wild Mammals: An Update. Viruses. 2019;11(5):459.
6. Saxena V, Bolling BG, Want T. West Nile Virus. Clinics in Laboratory Medicine. 2017;37(2):243-252.
7. Schaefer A, Js R, Goldstein J, Ryan C, Fair P, Gd B: Serological Evidence of Exposure to Selected Viral, Bacterial, and Protozoal Pathogens in Free-Ranging Atlantic Bottlenose Dolphins (Tursiops truncatus) from the Indian River Lagoon, Florida, and Charleston, South Carolina. Aquatic Mammals 2009:35:163-170.
8. St Leger J, Wu G, Anderson M, Dalton L, Nilson E, Wang D: West Nile virus infection in killer whale, Texas, USA, 2007. Emerg Infect Dis 2011:17(8):1531-1533.
9. Vandevelde, Marc, Robert Higgins, and Anna Oevermann. Veterinary neuropathology: essentials of theory and practice. John Wiley & Sons, 2012:56-57