Conference 19, Case 3:
Signalment:
A 2-year-old female spayed Pitbull (Canis lupus familiaris)
History:
A two-year old pit bull was hospitalized for management of acute liver injury and coagulopathy two days after ingesting three bulbs of cycad palm. Despite aggressive treatment, the patient developed hepatic encephalopathy and shock. Due to a poor prognosis, humane euthanasia was elected.
Gross Pathology:
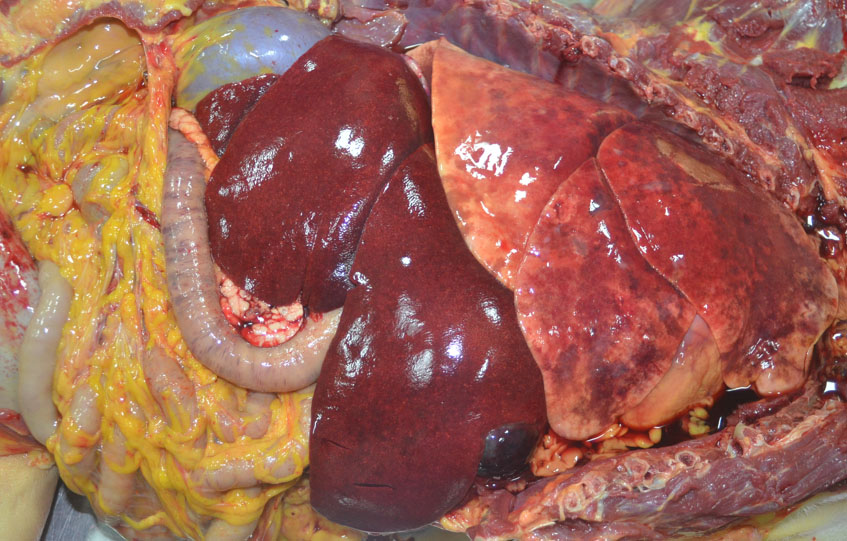
There was generalized icterus including the skin, mucus membranes, subcutaneous and mesenteric adipose tissue, fascia, and sclera, as well as petechiae and ecchymoses. The liver was enlarged, dark red and granular. There were multiple gastric mucosal ulcers, and melena throughout the gastrointestinal tract.
Laboratory Results:
The dog had hypoalbuminemia, hypocholesteremia, and hypoglycemia as well as unmeasurable PT and PTT, and severe thrombocytopenia. ALT, AST, GGT, ALP, and total bilirubin were all elevated.
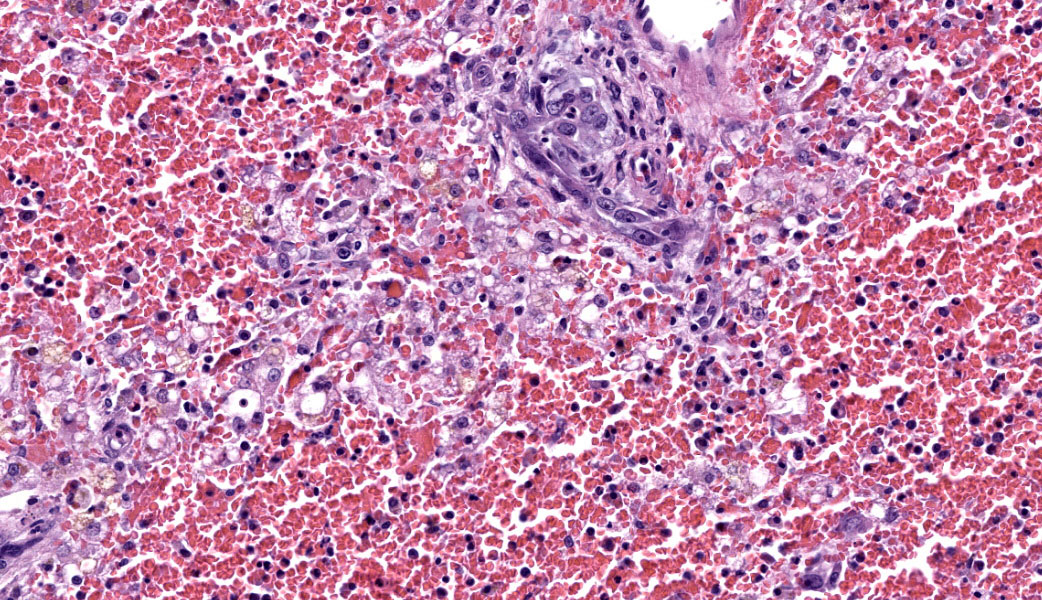
Microscopic Description:
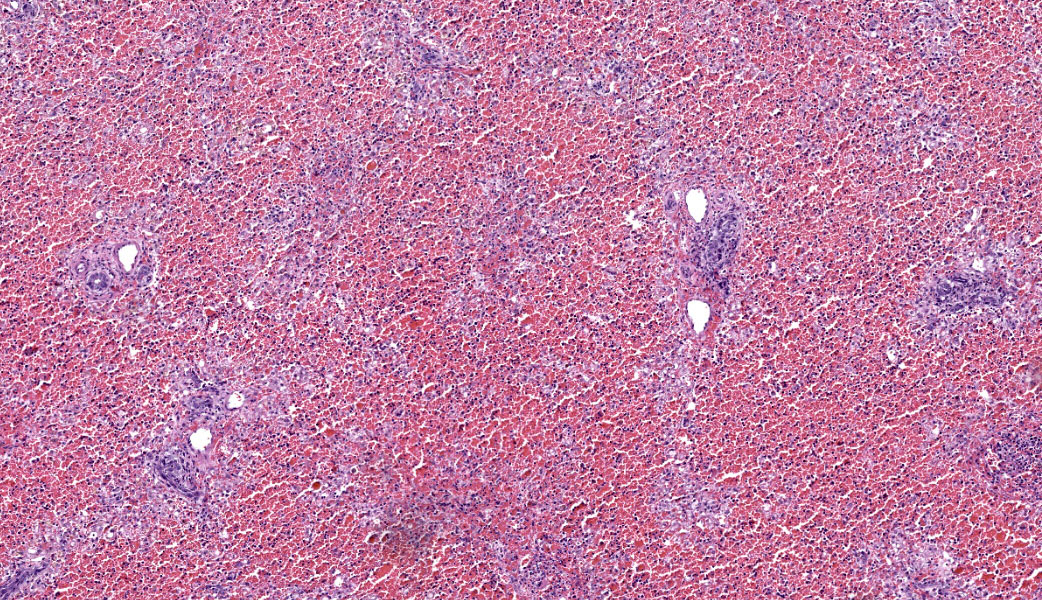
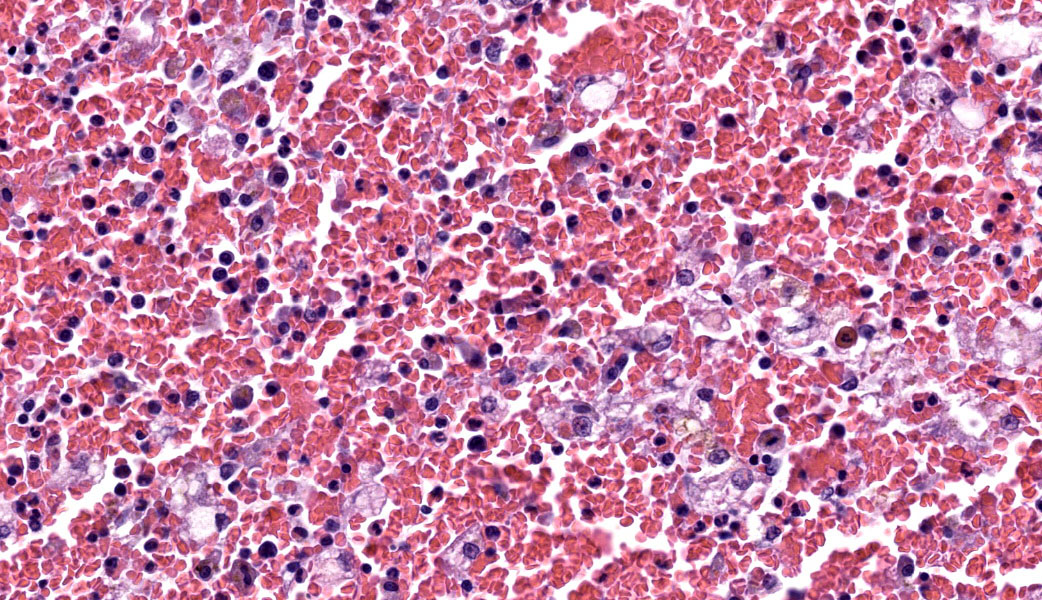
Liver: There is massive loss of hepatocytes in all examined sections with approximately 99% of the hepatic parenchyma affected. The portal regions contain scant numbers of remaining hepatocytes with large numbers of macrovesicular and microvesicular lipid vacuoles. Kupffer cells and occasional hepatocytes contain moderate amounts of yellow brown pigment (lipofuscin). There is moderate proliferation of oval cells, which have large open nuclei with a prominent nucleolus. There are increased numbers of circulating neutrophils and monocytes within the sinusoids.
Contributor’s Comment:
Cycad palms are primitive palm-like plants that are native to tropical and subtropical regions of the world. All cycads belong to the class Cycadopsida and the order Cycadales. In the United States common cycads palms include Dycas revoluta, Cycas circinalis, and Zaminia floridana and are commonly referred to as sago palms. In addition to being native in some regions, they are also popular ornamental plants in non-native climates.1,5 All parts of the cycad plant are toxic but the highest concentration of toxin is found in the seeds.11
Cycads contain three types of toxins: azoxyglycosides, of which cycasin is the most prominent member, b-methylamino-L-alanine (BMAA), and an unidentified high molecular weight compound.1,11 Cycasin, the predominant toxin, is metabolized by intestinal bacteria into the toxic metabolite methylazoxymethanol (MAM). MAM is neurotoxic, carcinogenic, mutagenic, teratogenic, and hepatotoxic.1,5,11 This carcinogen potential has been documented in rats where chronic exposure leads to cancer development in the liver, kidney, and intestinal tract of laboratory rodents and nonhuman primates.8,14 Once metabolized in the intestine, MAM undergoes enterohepatic circulation and enters the liver via the portal vein where it is metabolized by hepatic cytochrome P450 (CYPs).
Cycad ingestion in dogs leads to gastrointestinal and hepatic disease caused by methylazoxymethanol (MAM). Common clinical signs in dogs include vomiting, diarrhea occasionally with melena, abdominal pain, anorexia, icterus, hypersalivation, depression, and neurological signs secondary to hepatic encephalopathy. Common clinical pathology findings include hyperbilirubinemia, elevated alanine aminotransferase (ALT), elevated alkaline phosphatase, hypocholesterolemia, hypoalbuminemia, elevated prothrombin time (PT), elevated partial thromboplastin time (PTT), and thrombocytopenia.1,2,5,11,13
There are multiple retrospective studies on cycad poisoning in dogs with survival rates ranging from 32 to 64%.1,3,5 Severe liver injury leads to decreased synthesis of coagulation factors, decreased production of albumin, and disrupted bile acid circulation leading to decreased intestinal absorption of vitamin K.5,11 All of these lead to the observed clinical abnormalities. Increased vascular permeability also leads to albumin loss through intestinal and renal losses. Thrombocytopenia may occur due to coagulopathy and gastrointestinal bleeding.
MAM is oxidized by hepatic cytochrome P450 (CYPs) leading to centrilobular necrosis as the most common finding in acute toxicosis. Subacute changes include severe centrilobular coagulative necrosis with some regeneration as well as portal fibrosis and bile duct proliferation. Chronic changes include diffuse hepatocellular necrosis, hepatic degeneration and regeneration with extensive stromal collapse, bridging fibrosis with nodular regeneration, and biliary hyperplasia.5 There are multiple retrospective studies on cycad poisoning in dogs with survival rates ranging from 32 to 64% and even with recovery chronic hepatic disease can persist.1,3,5
Cycad intoxication also occurs in domestic ruminants where it manifests as two distinct syndromes: neurologic and hepato-gastrointestinal.6,7,8,12 The hepatic-gastrointestinal syndrome causes hepatocellular megalocytosis, centrilobular necrosis, and biliary duct hyperplasia. These may be similar to other hepatic toxicoses such as aflatoxin, pyrrolizidine alkaloids, Lantana species, and spordesmin. As in dogs, cycasin and its metabolites have been linked to hepatic-gastrointestinal syndrome in ruminants, and the hepato-gastrointestinal syndrome is more common in sheep compared to the neurologic syndrome which is more common in cattle.1
Cycads also contain the neurotoxin beta-methylamino-L-alanine (BMAA) and an unidentified neurotoxin. This unidentified neurotoxin is thought to be the cause of neurologic disease in cattle with chronic cycad poisoning, where progressive proprioception deficits in the hindlegs are attributed to axonal degeneration in the dorsal funiculus and the spinocerebellar and corticospinal tracts.7,8,12
Cycad intoxication is associated with severe disease and fatalities in multiple domestic species caused primarily by hepatotoxicity which leads to secondary effects of hepatic failure including hepatic encephalopathy, coagulopathy, and hypoproteinemia.1-5,11,13 In the cattle, cycad intoxication can also cause neurologic disease due to axonal degeneration.7,8,12
Contributing Institution:
Louisiana Animal Disease Diagnostic Laboratory (LADDL)
School of Veterinary Medicine, Louisiana State University
(http://www1.vetmed.lsu.edu/laddl/index.html)
JPC Diagnoses:
Liver: Hepatocellular necrosis, acute, massive, diffuse, with hemorrhage and stromal collapse.
JPC Comment:
This contributor also provides an outstanding comment on this entity, which represents a case of acute hepatic necrosis secondary to a toxin in a dog. Much of what was discussed in conference for this case is covered in their write-up. Hepatic damage by cycad palm is progressive and dose-dependent.9 Dr. Whitten discussed the acute changes seen in toxicity cases such as this, which include centrilobular hemorrhage & massive hepatocellular coagulative necrosis, vacuolar degeneration, hepatic venous congestion, pigmentary bile duct distention, bile duct plugging, and intracanalicular cholestasis.
Participants were also asked about various other potential causes of of massive hepatic necrosis across numerous species. Other toxic and chemical causes discussed included heavy metals, such as copper toxicity in sheep and young cattle, arsenic, and iron; medications/drugs, with specific mention of high-dose ivermectin, some antibiotics, halothane, and phenobarbital; plants, fungi, and algae, such as Amanitaspp. toadstools, Senecio spp. and Crotalaria spp. (pyrrolizidine alkaloids), Aspergillusspp. (aflatoxins), Fusarium spp. (fumonisins), and blue-green algae (cyanobacteria); and industrial chemicals like carbon tetrachloride, chlorinated hydrocarbons, and pesticides (i.e., paraquat).4 Infectious causes included canine adenovirus-1 (canine infectious hepatitis), canine herpesvirus-1, rabbit calicivirus (rabbit hemorrhagic disease virus), Clostridium novyi (Black Disease), and Salmonella spp. A variety of ischemic and metabolic causes were mentioned and, in summary, covered shock/hypoxia, thromboembolisms, heat stroke, and DIC.4 A brief mention of liver flukes, such as Fasciola hepatica, was made due to their contribution to the development of Black Disease via creation of migration tracks that enable a hypoxic environment for Clostridium novyi to proliferate. Lastly, there was brief discussion on the massive hepatic necrosis seen with Theiler’s disease in horses (caused by equine parvovirus) and the lesions of inherited copper storage disease in Bedlington Terrier and Doberman dogs.
In cases of cycad palm in dogs, cattle, and rodents, lesions of the CNS have been reported in addition to the gastro-hepatic syndrome.6,7,8,12 The primary lesions seen in the CNS include vacuolation of the neuropil, demyelination, and spongiform change to the brain and spinal cord.8,12 While sago palm is known to produce BMAA neurotoxin, Dr. Whitten speculated that some of the brain lesions (i.e., astrocyte swelling and cytotoxic edema) may also be secondary to hepatic encephalopathy from severe hepatic necrosis.
References:
- Albretsen JC, Khan SA, Richardson JA. Cycad palm toxicosis in dogs: 60 cases (1987-1997). J Am Vet Med Assoc. 1998;213: 99-101.
- Botha CJ, Naude TW, Swan GE, Ashton MM, van der Wateren JF. Suspected cycad (Cycas revoluta) intoxication in dogs. J S Afr Vet Assoc. 1991;62: 189-190.
- Clarke C, Burney D. Cycad Palm Toxicosis in 14 Dogs from Texas. J Am Anim Hosp Assoc. 2017;53: 159-166.
- Cullen JM, Stalker MJ. Liver and Biliary System. In: Maxie MG, ed. Jubb, Kennedy & Palmer's Pathology of Domestic Animals. Vol 2. 6th ed. St. Louis, MO: Elsevier; 2016:258-352.
- Ferguson D, Crowe M, McLaughlin L, Gaschen F. Survival and prognostic indicators for cycad intoxication in dogs. Journal of veterinary internal medicine. 2011;25: 831-837.
- Gabbedy BJ, Meyer EP, Dickson J. Zamia palm (Macrozamia reidlei) poisoning of sheep. Aust Vet J. 1975;51: 303-305.
- Hall WT. Cycad (zamia) poisoning in Australia. Aust Vet J. 1987;64: 149-151.
- Hooper PT, Best SM, Campbell A. Axonal dystrophy in the spinal cords of cattle consuming the cycad palm, Cycas media. Aust Vet J. 1974;50: 146-149.
- Lake BB, Edwards T, Atiee G, Stone R, Scott L. The characterization of cycad palm toxicosis and treatment effects in 130 dogs. Aust Vet J. 2020;98(11):555-562.
- Laqueur GL, Matsumoto H. Neoplasms in female Fischer rats following intraperitoneal injection of methylazoxy-methanol. J Natl Cancer Inst. 1966;37: 217-232.
- Milewski LM, Khan SA. An overview of potentially life?threatening poisonous plants in dogs and cats. Journal of Veterinary Emergency and Critical Care. 2006;16:25-33.
- Reams RY, Janovitz EB, Robinson FR, Sullivan JM, Rivera Casanova C, Mas E. Cycad (Zamia puertoriquensis) toxicosis in a group of dairy heifers in Puerto Rico. J Vet Diagn Invest. 1993;5:488-494.
- Senior D, Sundlof S, Buergelt C, Hines S, O'Neil-Foil C, Meyer D. Cycad intoxication in the dog. The Journal of the American Animal Hospital Association (USA). 1985.
- Sieber SM, Correa P, Dalgard DW, McIntire KR, Adamson RH. Carcinogenicity and hepatotoxicity of cycasin and its aglycone methylazoxymethanol acetate in nonhuman primates. Journal of the National Cancer Institute. 1980;65: 177-189.