Cofnerence 19, Case 2
Signalment:
7.5-year-old male cynomolgus macaque (Macaca fasicularis)
History:
This animal had undergone surgery six days previously to induce acute hepatic failure by ischemic liver injury and had concurrently received a xenographic transplant of swine hepatocytes into the spleen. It had been started on tacrolimus immunosuppressive therapy prior to manipulation. Two days post-operatively, the monkey began progressive clinical deterioration as well as developing diabetes. It was unresponsive to empirical supportive therapy and was euthanized.
Gross Pathology:
Kidneys were reported to be pale and reduced in size without additional specifications.
Laboratory Results:
N/A.
Microscopic Description:
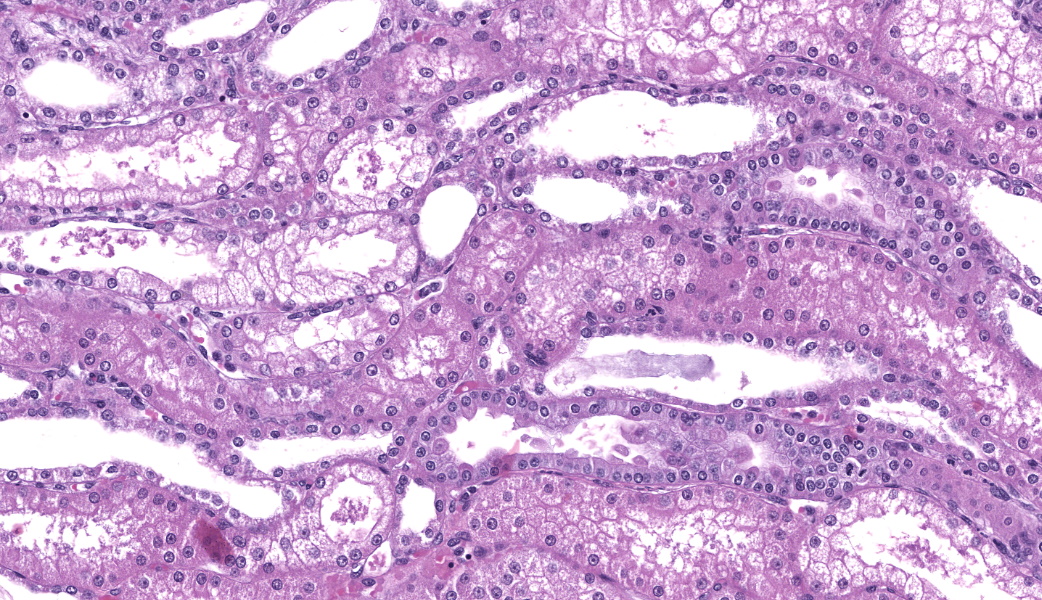
Renal lesions involve primarily the tubular component and are characterized by a variety of morphological features. Swelling and vacuolar change are prominent within proximal renal tubular epithelium, especially in the straight portion of that segment. Many tubules contain prominent cast-structures, typically consisting of granular to globular eosinophilic material, frequently admixed with blueish-gray proteinaceous sheets. Exfoliated epithelial cells are occasionally incorporated into the cast structures with numerous tubules also containing aggregates of mixed, primarily granulocytic inflammatory cells. Individually necrotic renal tubular epithelium is occasional evident. Associated with obstruction created by these casts, there is extensive, widespread tubular dilatation. Although extremely infrequent, fibrin thrombi were noted within occasional glomerular capillary loops. However, other significant glomerular changes were uncommon at the light microscopic level.
Contributor's Morphologic Diagnoses:
1. Renal tubular vacuolization, patchy to focally extensive, primarily proximal tubules, moderate-marked.
2. Coarsely granular eosinophilic tubular casts with occasional proteinaceous sheets consistent with myoglobin and associated myoglobinuric nephrosis, patchy, moderate, with associated marked tubular dilatation and epithelial cell thinning.
3. Acute tubular injury (ATI), patchy, mild, characterized by renal tubular epithelial cell exfoliation, individual cellular degeneration and patchy, primarily granulocytic inflammatory cell aggregates.
4. Focal glomerular capillary loop fibrin thrombi, minimal.
Contributor's Comment:
Despite administration at standard dosing levels, this animal's tacrolimus levels climbed at one point to 80 ng/ml (target range <16 ng/ml) and the monkey developed clinical signs of toxicity as well as diabetes. Unfortunately, renal function values were not available at the time of necropsy.
Tacrolimus, also known as FK506 is a macrolide immunosuppressive drug derived from the fungus Streptomyces tsukubaensis, approved by the FDA in 1994 to lower the risk of allogenic organ transplant rejection. Although not sharing molecular structural properties, it has similar therapeutic mechanisms and toxicity with its older immunosuppressive counterpart, cyclosporine. Both are classified as calcineurin inhibitors (CNI), affecting calcium-dependent events such as nitric oxide synthase activities, apoptosis, and cell degranulation.13
Renal toxicity is a well-recognized effect, both to transplanted kidneys as well as the native kidneys of patients receiving other organs. Early nephrotoxicity is primarily associated with acute arteriole vasoconstriction and is thought to be of multifactorial pathogenesis, including increasing vasoconstrictive factors, reducing vasodilator mechanisms and increasing the formation of free radicles.3,4,6,7,14 Toxicity due to vasospasms can manifest in a so-called "functional form" without any characteristic morphological changes, or with associated structural histological alterations, either
acute or chronic.13 The latter includes effects on both arterioles (especially afferent arterioles) and glomeruli, including smooth muscle cell swelling, endothelial cell injury/necrosis and fibrin thrombi, potentially resulting in thrombotic microangiopathy. Such changes were considered to be relatively modest in this case, although very mild mesangial matrix expansion and hypercellularity of the juxtaglomerular apparatus were occasionally noted. Tubular components of CNI injury can include the development of vacuolization leading to clear, aqueous fluid accumulation (the latter being associated ultrastructurally with dilated smooth endoplasmic reticulum and occur predominantly in the straight portion of the proximal tubules.) This process was prominent in this monkey. As vacuolar change is a common, non-specific lesion often seen with osmotic nephrosis due to agents such as mannitol, inulin, glucose, radiocontrast agents, chemicals that induce vacuolation through phagolysosomes and other causes of tubular ischemia correlation with other factors must be taken in ascribing its pathogenesis.7
Another form of renal tubular injury is occurring in this case - likely tacrolimus related as well, but of a more secondary mechanism. Since early after its therapeutic use, tacrolimus has been known to be associated with severe rhabdomyolysis, although the incidence is considered rare.1,2 The role of calcineurin in skeletal muscles is complex, with both protective and detrimental effects.14 CNI are potent CYP450 inhibitors and this may be related to an increased incidence of muscle toxicity in transplant recipients, especially associated with other drug interactions.1
Renal casts noted in this case (and often admixed with numerous dehisced epithelial cells) were suggestive of myoglobin origin. They are often comprised of round granules that line up in chains or aggregate in clusters. Tinctorially, there is variation between and even within different stains applied with a typical pink to red-brown appearance with H&E, pink to bright magenta with PAS, and often a gray-blue sheet containing fine red granules on Trichrome.5,8 Immunostaining with antibody to myoglobin was strongly positive in these casts, often with a prominent rope-like string to beaded appearance. Interestingly, the concept that acute kidney injury is triggered by myoglobin as a direct, sole toxin appears to be oversimplified and there is ample evidence that additional factors such as hypovolemia, renal vasoconstriction or urine acidification are required for nephrotoxicity.9,12 Of these factors, certainly vasoconstriction directly associated with CNI effects would have likely been present. Again, in this case the myoglobinuric nephrosis present cannot be definitively ascribed to tacrolimus, but this is considered a likely pathogenesis, especially in the absence of other common pathogeneses such as severe trauma, dehydration, and other frequently associated comorbidities.5
Although both the direct and indirect effects of tacrolimus toxicity in this case were thought to be substantially responsible for lesions, the impact of the severe ischemic hepatic and biliary injury created prior to the transplant was also considered to have potentially played a role in some of the changes present.
Hepatorenal syndrome (HRS) involves the development of renal failure in patients with severe liver disease and while often seen in chronic cirrhosis, it can also occur in patients with acute liver disease.10 The pathogenesis is associated with severe renal vasoconstriction resulting from complex changes in splanchnic and general circulations as well as systemic and renal vasoconstrictors and vasodilators.10 This entity is mentioned only to emphasize the complexity and interconnectedness of the kidneys with other systemic functions.
In addition to renal effects, other common toxic complications associated with tacrolimus use in humans include neurological problems and diabetes, although numerous other clinical problems such as diarrhea, pruritus and alopecia are reported.16
Contributing Institution:
Division of Laboratory Animal Resources, University of Pittsburgh; www.dlar.pitt.edu
JPC Diagnoses:
Kidney: Tubular vacuolization, necrosis, and regeneration, acute, multifocal, marked, with granular casts and glomerular fibrin thrombi.
JPC Comment:
The contributor of this case provided an exceptional comment and covered much of what was discussed in conference. Hemoglobin and myoglobin were discussed as the two main rule-outs for the granular casts present in the kidney and special attention was drawn to the fact that, with tacrolimus toxicity, the vacuolation of the straight portion of the proximal convoluted tubules (PCT) is the primary process and is associated with dilation of the smooth endoplasmic reticulum of the PCT epithelial cells.14 This kidney presented a couple of nice examples of tubular regeneration as well. Additional ruleouts in this case that were considered were aminoglycoside toxicity, which tends to appear more like a lysosomal storage disease with build-up of cytoplasmic material in the PCT epithelial cells, mannitol administration, which can cause osmotic nephrosis when given in high volume or over a prolonged period of time, and any other toxins that inhibit CYP450 that may have also resulted in tubular epithelial vacuolation.
CYP450 enzymes in the kidney are critical for maintaining fluid and electrolyte balance, regulating blood pressure, and metabolizing drugs or toxins. They function primarily in the PCTs to catalyze the oxidation of arachidonic acid into vasoactive substances, such as certain HETE molecules (vasoconstrictors) and epoxyeicosatrienoic acids (EETs; vasodilators), which all help to modulate vascular tone and tubular sodium transport.11 These potent signaling molecules act as protective, locally acting agents in the kidney to reduce blood pressure, decrease inflammation, and prevent tissue fibrosis.11 When CYP450 is inhibited, these molecules are not produced effectively, resulting in kidney injury.
References:
- Dopazo C, Bilbao I, Lazaro JL et al. Severe Rhabdomyolysis and Acute Renal Failure Secondary to Concomitant Use of Simvastatin With Rapamycin Plus Tacrolimus in Liver Transplant Patient. Transplantation Proceedings. 2009;41:1021-1024.
- Hibi S, Misawa A, Tsunamoto K et al. Severe rhabdomyolysis associated with tacrolimus. Lancet. 1995;346(8976):70.
- Issa N, Kukla A and Ibrahim HN. Calcineurin Inhibitor Nephrotoxicity: A Review and Perspective of the Evidence. Am J Nephrolo. 2013;37:602-612.
- Kemper J and Kniska K. Pathophysiology and treatment of calcineurin inhibitor nephrotoxicity. Kidney Centric. 2014;1-6.
- Liapis H, Boils CB, Hennigar R and Silva F. Myoglobin casts in renal biopsies: immunohistochemistry and morphologic spectrum. Human Pathology. 2016;54:25-30.
- Myers BD. Cyclosporine nephrotoxicity. Kidney International. 1986;30:964-974.
- Naesens M, Kuypers DR and Sarwal M. Calcineurin Inhibitor Nephrotoxicity. American Society of Nephrology. 2009;4:481-508.
- Najafian B, Fogo AB, Lusco MA and Alpers CE. AJKD Atlas of Renal Pathology: Myoglobin Cast Nephropathy. Am J Kidney Dis. 2017;69(2):e7-e8.
- Najafian B, Frankinn DB and Fogo AB. Acute Renal Failure and Myalgia in a Transplant Patient. J Am Soc Nephrol. 2007;18:2870-2874.
- Ng C, Chan M, Morris HI et al. Hepatorenal Syndrome. Clin Biochem Rev. 2007;28:11-17.
- Omata K, Abe K, Sheu HL, et al. Roles of renal cytochrome P450-dependent arachidonic acid metabolites in hypertension. Tohoku J Exp Med. 1992;166(1):93-106.
- Petejova N and Martinek A. Acute kidney injury due to rhabdomyolysis and renal replacement therapy: a critical review. Critical Care. 2014;18:224.
- Renal Transplantation Pathology. In: Jennette JC, Olson JL, Silva FG and D'Agati VD eds. Hepinstall's Pathology of the Kidney Vol II. 7th Philadelphia US: Wolters Kluwer; 2015:1321-1460.
- Randhawa PS, Starzl TE and Demetris AJ. Tacrolimus (FK506)-Associated Renal Pathology. Adv Anal Pathol. 1997;4(4):265-276.
- Sathyan S, Baskharoun R and Perlman AS. Prevention of Recurrent Episodes of Rhabdomyolysis With Tacrolimus in a Transplant Recipient With Myopathy. American Journal of Therapeutics. 2014;21:e171-e174.
- Scott LJ, McKeage K, Keam SJ, et al. Tacrolimus: A further update of its use in the management of organ transplantation. Drugs. 2003;63:1247-1297