CASE II:
Signalment:
Adult male Fischer 344 (F344) rat (Rattus norvegicus)
History:
This rat was part of a research study and was exposed to a chemical on 10/20/2014. He was then found dead on 11/28/2024.
Gross Pathology:
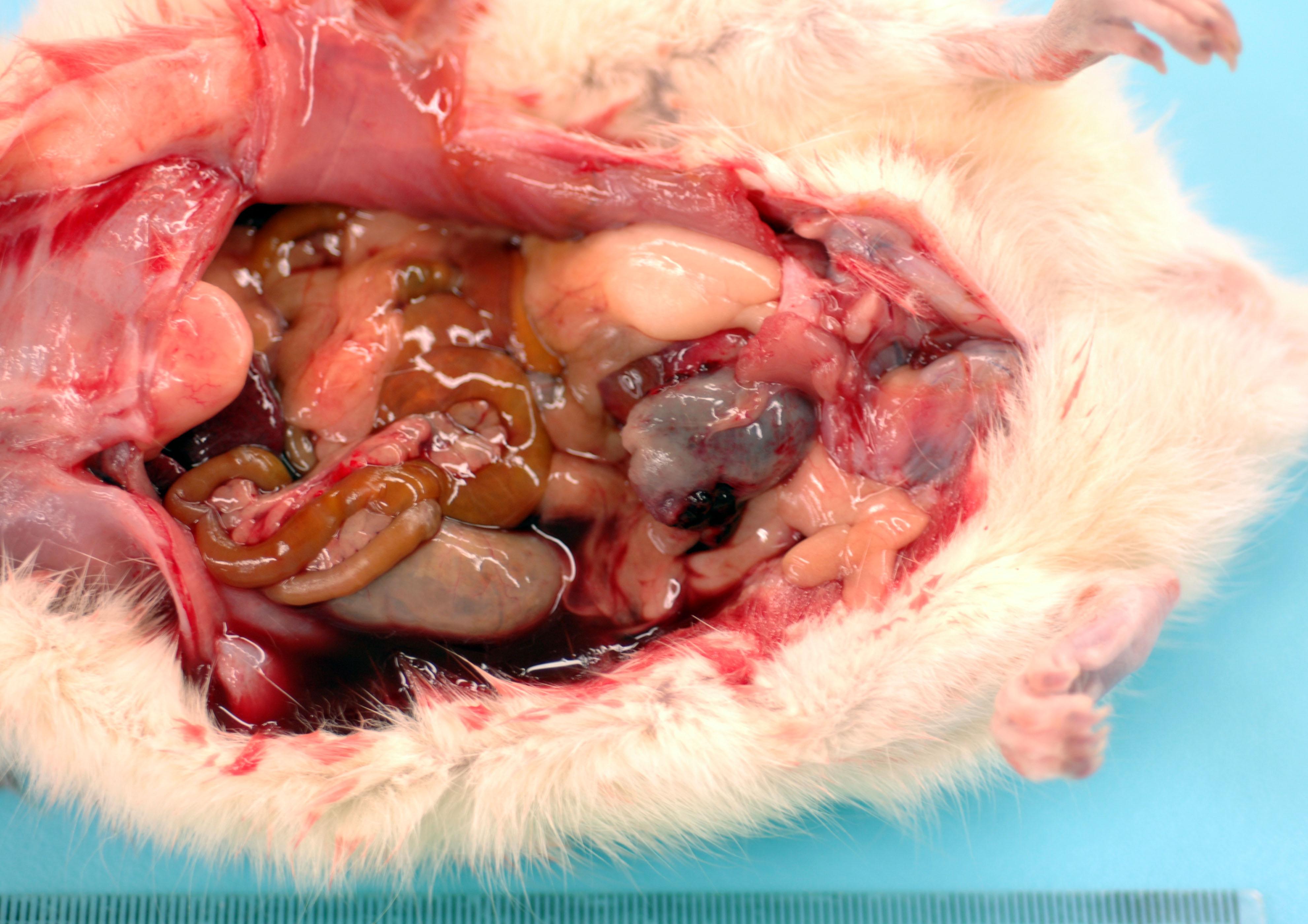
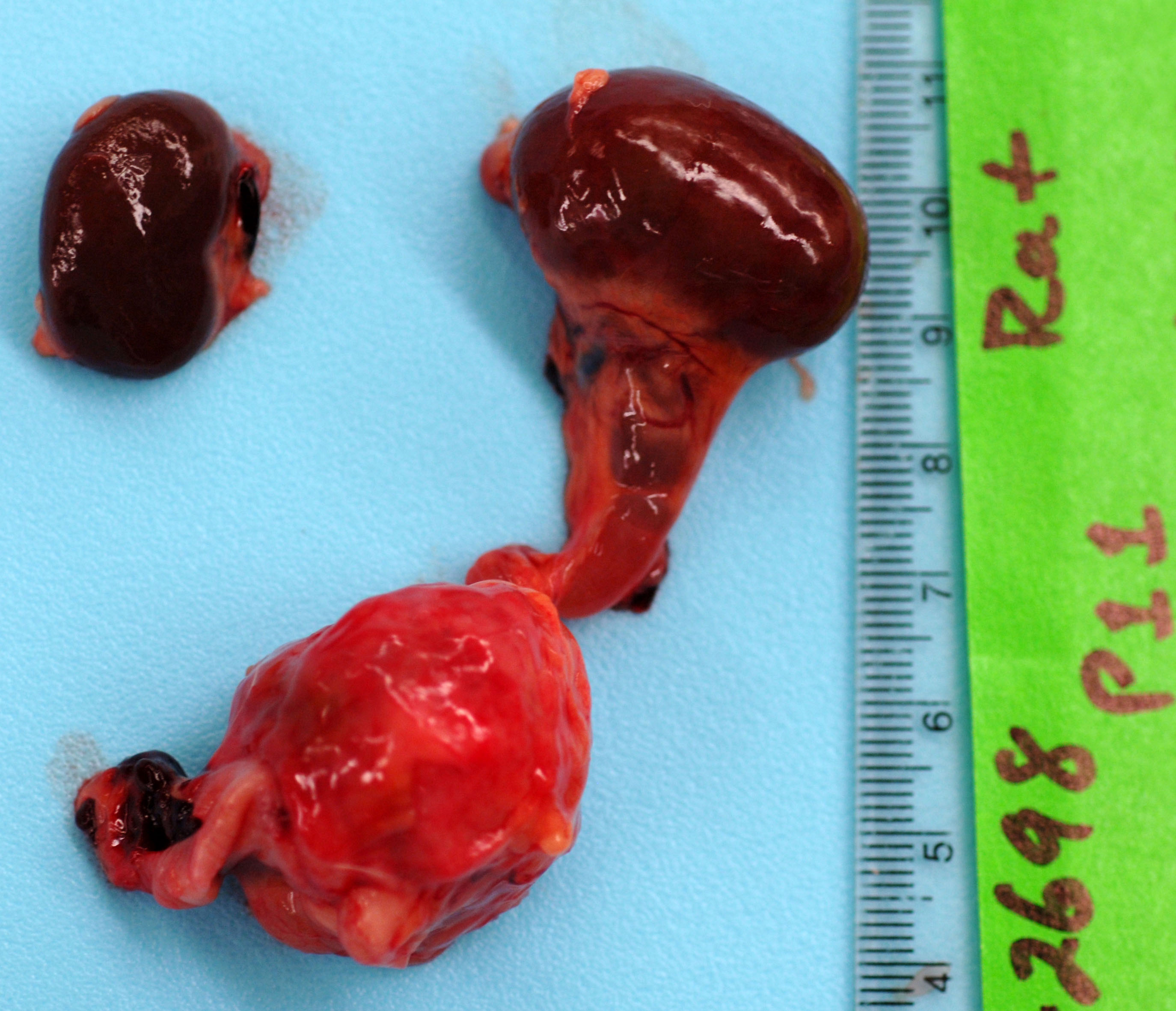
Received for autopsy is a 389g, adult, male Fischer 344 rat with a body condition score of 3/5, adequate subcutaneous and visceral fat, and in full rigor mortis. There is mild autolysis. There is multifocal, patchy thinning of the haircoat along the dorsum with multifocal scabs; a section is taken for histology. The abdominal cavity feels doughy. Upon incising the abdominal cavity, there is approximately 10 mL of serosanguineous fluid in the abdomen (uroperitoneum). The lungs collapse when the diaphragm is cut; they are inflated with 10 mL of 10% NBF. The stomach is full of yellow-gray ingesta. There is scant to moderate amounts of green-brown digesta in the moderately autolyzed small intestines. The cecum is full of dry, green-gray digesta. The distal colon in empty and the proximal colon contains few fecal pellets. The urinary bladder is ruptured with clotted blood adhered to the edges of the rupture. On the dorsal left side of the urinary bladder is a 2.5 cm, round, firm, mass that is pale white and homogenous with central necrosis on cut section. The left kidney is 3 x 2 x 1.5 cm and the left ureter is 5 mm in diameter (hydronephrosis and hydroureter). There are multifocal red-orange, 1-3 mm foci of neoplasia in the right testicle (suspect interstitial cell tumor). All other organs examined appear grossly normal.
Laboratory Results:
Cytologic findings (touch impression of the mass): This fair quality, highly cellular cytologic specimen is composed of a mixture of rafts of tightly adherent polygonal epithelial cells, smaller clusters of epithelial cells, and occasional individualized epithelial cells. There is marked anisocytosis and anisokaryosis with cells measuring 3-12 erythrocytes in diameter and nuclei measuring 2-8 erythrocytes in diameter. Cells have abundant light blue, occasionally vacuolated cytoplasm. Nuclei have reticulated chromatin and occasional prominent nucleoli. Many cells exhibit high nuclear:cytoplasm ratio. There are occasional bi- and multi-nucleated cells. This is on a background of abundant cellular debris, viable and degenerate neutrophils, cocci and coccobacilli bacteria, macrophages, stain precipitate, and naked and streaming nuclei.
Microscopic Description:
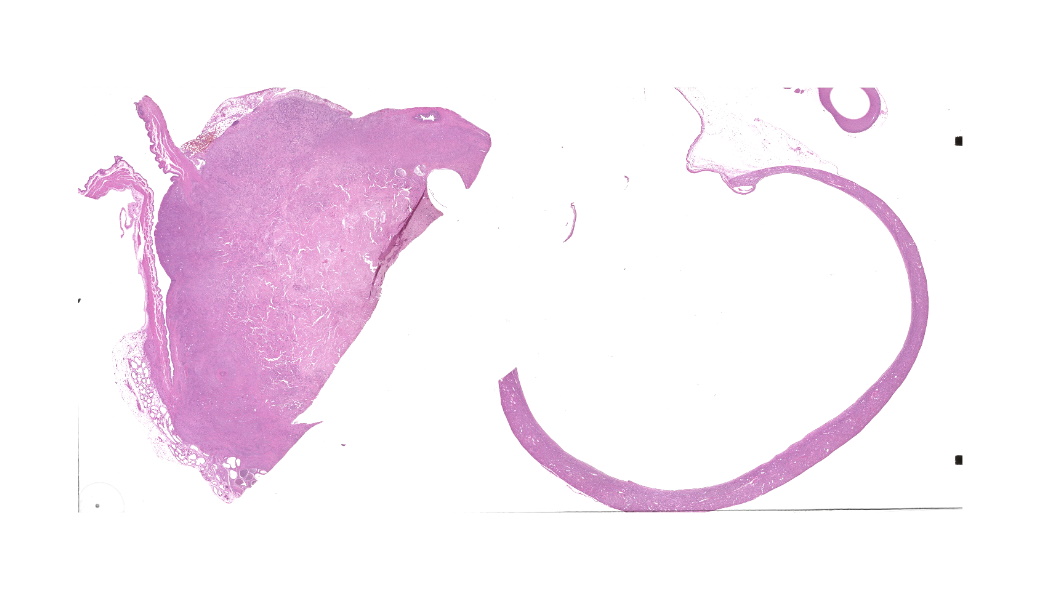
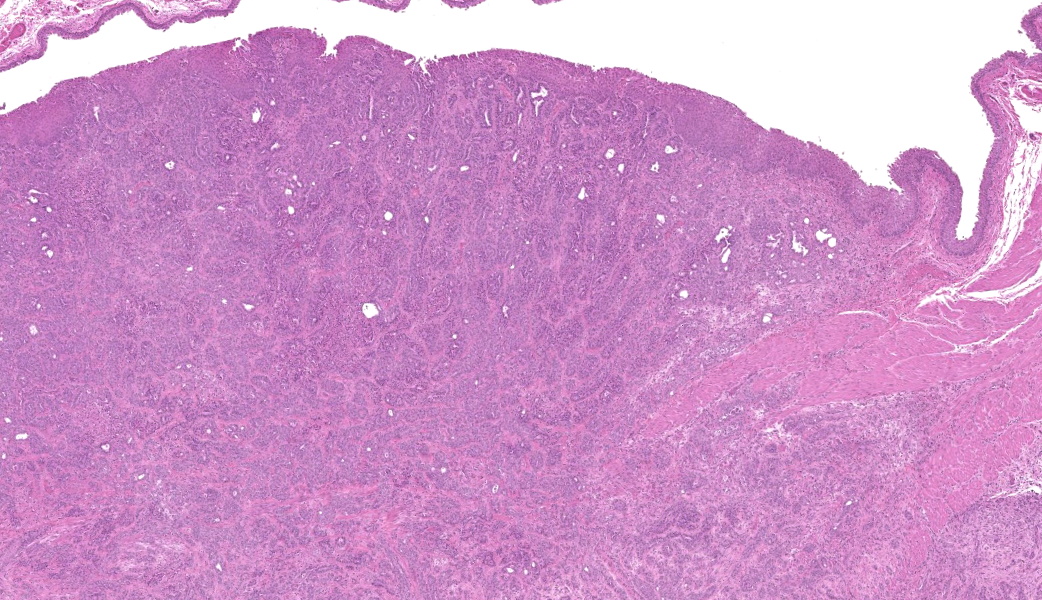
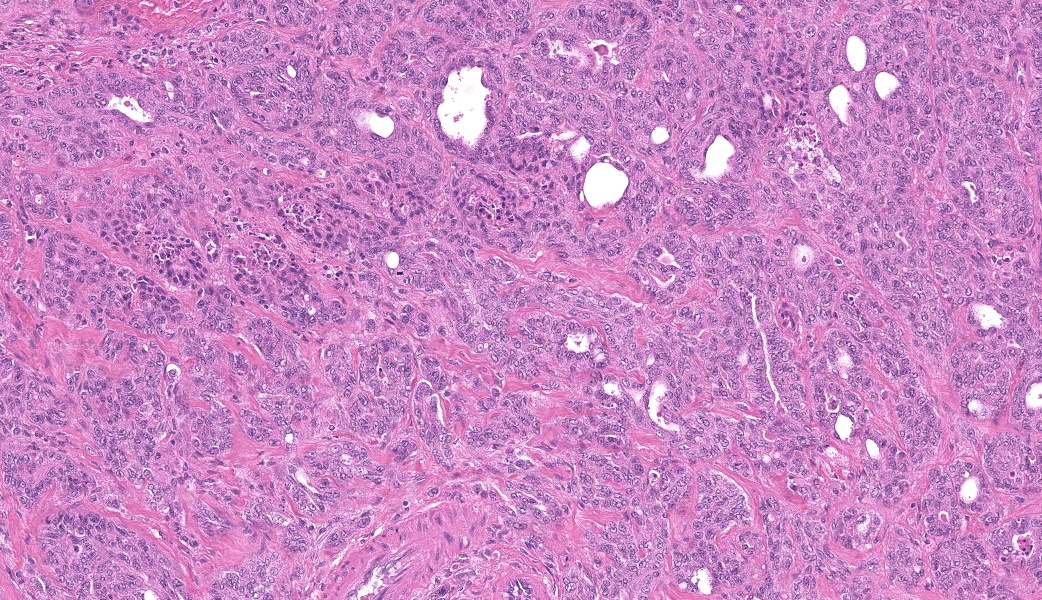
Urinary bladder: Arising from urothelium, infiltrating and expanding the lamina propria, and surrounding the ureter is a non-
encapsulated, well demarcated, moderately cellular neoplasm composed of polygonal cells arranged in nests on a fibrovascular stroma. Each neoplastic cell has indistinct cell borders, a moderate amount of pale basophilic cytoplasm, with a single misshapen nucleus with finely stippled chromatin and 1-3 distinct nucleoli. Anisocytosis and anisokaryosis are moderate. There are 2 mitotic figures per 2.37mm2. The neoplastic nests are often separated by a robust scirrhous response. Multifocally the neoplastic cells are replaced by eosinophilic, amorphous material (necrosis).
Prostate gland: Multifocally, prostatic glands are filled with cellular debris and degenerate neutrophils.
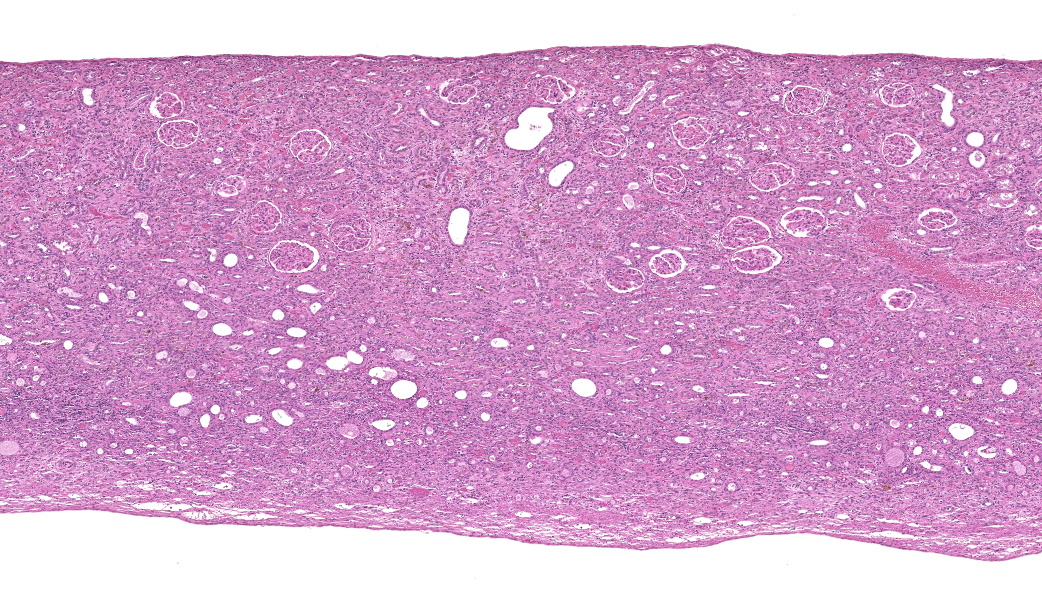
Kidney: There is severe dilation of the renal pelvis with marked thinning of the renal medulla and cortex.
Contributor's Morphologic Diagnosis:
Urinary bladder: Urothelial cell carcinoma, Rattus norvegicus.
Kidney: Hydronephrosis, severe.
Prostate gland: Prostatitis, neutrophilic, mild.
Contributor's Comment:
The cause of death in this animal is a ruptured urinary bladder secondary to outflow obstruction from a urinary tumor. The tumor observed both grossly and microscopically is consistent with a urothelial cell carcinoma of the urinary bladder epithelium. Spontaneous urothelial cell carcinomas of the urinary bladder are rare in rats. Although they may be locally invasive, they rarely metastasize. In this case, both the size of the tumor and the strong scirrhous response contributed to the outflow tract obstruction (and therefore an inability to urinate) and urinary inflow tract obstruction from the left kidney (resulting in unilateral hydronephrosis and hydroureter).
This case lacks Melamed-Wolinksi (MW) bodies, which are not listed as a feature in rats(1). MW bodies were first described in 1961 by Melamed and Wolinska while studying 500 urine cytology specimens(2) and are defined as single to many round red-or green cytoplasmic inclusions seen in degenerated urothelial cells; however, MW bodies are not always round.(3) Cells with MW bodies when noted in body fluids (e.g., pleural fluid, ascitic fluid) signify urothelial origin and suggest metastasis. Recent studies suggest MW bodies are enlarged lysosomes occurring in degenerating urothelial cells.(2)
In addition to the urinary bladder tumor, this case provides a great example of what happens upstream of the urinary bladder when it is blocked. At www.medicinepoems.com, there is an 8-stanza ode to hydronephrosis. The second stanza reads as follows:
Hydronephrosis, the name it bears in pain,
A flood within the kidney's fragile frame,
Where once smooth flow of life's essential stream,
Is halted now, a blocked and troubled dream.
Contributing Institution:
Comparative Pathology Department at the Institute of Chemical Defense https://usamricd.health.mil/
JPC Morphologic Diagnosis:
- Urinary bladder: Urothelial cell carcinoma.
- Kidney: Hydronephrosis, chronic, diffuse, severe.
JPC Comment:
First of all, if you missed the poem above, go read it now. It is a blessing for the WSC to have been graced by an ode to hydronephrosis such as this.
This case generated rich discussion on the importance of strain-specific background pathology. As is true for most laboratory rat strains, the Fischer 344s come with genetic baggage. They are predisposed to large granular lymphocytic leukemia, testicular mesothelioma (typically the epithelioid variant), and testicular interstitial cell tumors, the last of which can induce hypercalcemia in this strain. While none of these were primary considerations in this case, the reminder served as a useful anchor for approaching pathology in this particular strain.
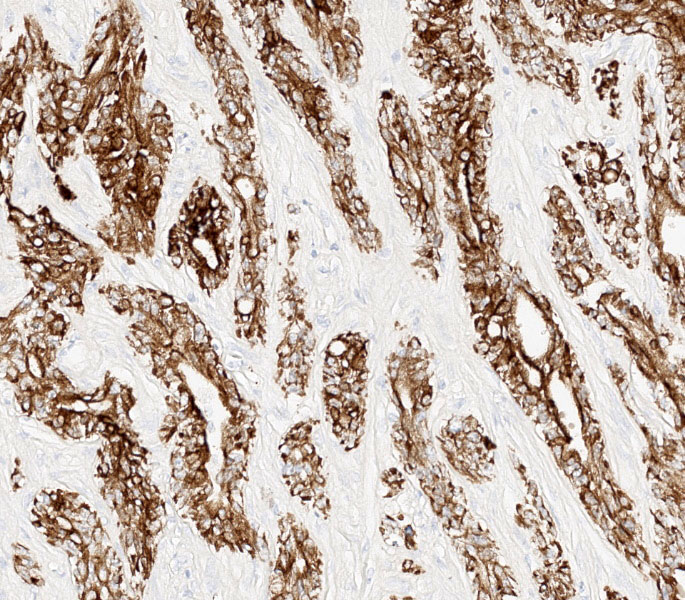
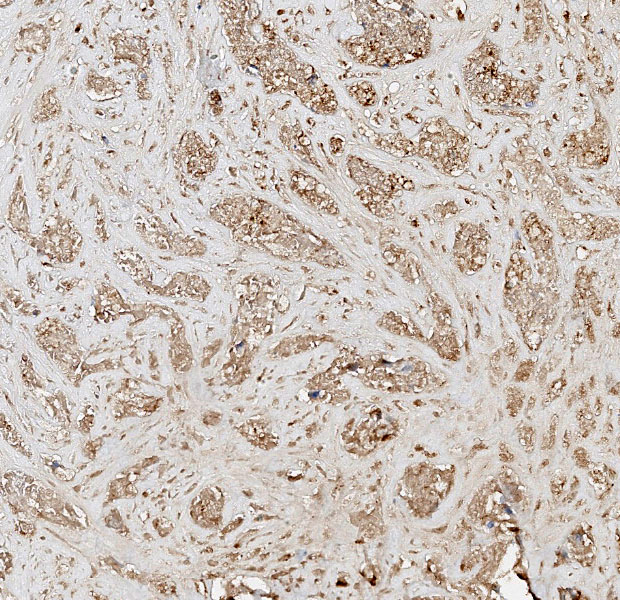
Differential diagnoses for this urinary bladder mass included urothelial cell carcinoma, prostatic carcinoma, and renal carcinoma, with the latter two included largely to ensure that metastatic or locally invasive disease was not being overlooked. The histologic architecture of an infiltrative epithelial neoplasm seemingly arising from the bladder wall favored a urothelial origin, but immunohistochemistry provided an additional layer of information. The neoplastic cells were diffusely immunoreactive for CK7, a cytokeratin expressed in most primary urinary bladder carcinomas.4 They were negative for GATA3, a transcription factor involved in urothelial differentiation, which may suggest prostatic carcinoma instead.5 However, Dr. Alves noted that GATA3 is most reliable in well-differentiated urothelial carcinomas, and its absence here is also consistent with less well-differentiated UCCs, as in this case.5 The tumor was also negative for PAX8, a marker of renal tubular epithelium, which helped exclude renal carcinoma.2 Uroplakin, a highly specific marker for urothelial cell carcinoma, unfortunately did not work in this case.7 Participants also discussed the glandular differentiation present within the tumor, which can be seen in both urothelial cell and prostatic carcinomas. In humans, glandular differentiation in urothelial carcinoma is associated with invasion and poorer differentiation.8 While extrapolating across species is something to be avoided, the same pattern was evident here.
One of the most instructive aspects of this case was the hydronephrosis, which was beautifully captured on the H&E slide. Dr. Alves emphasized not to overlook this lesion: the upstream consequences of urinary outflow obstruction (pelvic dilation, cortical and medullary thinning, tubular atrophy) were classic! The obstruction in this rat was likely caused by the combination of the tumor's size and its pronounced desmoplastic response, which occluded the bladder outlet.
References:
- Ayra P, Khalbuss WE, Monaco SE, Pantanowitz L. Melamed-Wolinska bodies. Diagn Cytopathol. 2012;40:150-1.
- Barr ML, Jilaveanu LB, Camp RL, Adeniran AJ, Kluger HM, Shuch B. PAX-8 expression in renal tumours and distant sites: a useful marker of primary and metastatic renal cell carcinoma?. J Clin Pathol. 2015;68(1):12-17.
- Frazier KS, Seely JC, Hard GC, et al. Proliferative and Nonproliferative Lesions of the Rat and Mouse Urinary System. Toxicologic Pathology. 2012;40(4_suppl):14S-86S.
- Jiang J, Ulbright TM, Younger C, et al. Cytokeratin 7 and cytokeratin 20 in primary urinary bladder carcinoma and matched lymph node metastasis. Arch Pathol Lab Med. 2001;125(7):921-923.
- Liang Y, Heitzman J, Kamat AM, Dinney CP, Czerniak B, Guo CC. Differential expression of GATA-3 in urothelial carcinoma variants. Hum Pathol. 2014;45(7):1466-1472.
- Rashidi B, Tongson-Ignacio JE. Melamed-Wolinska bodies in urine cytology an interesting aggregate in a degenerated urothelial cell. Diagn Cytopathol. 2011;39:117.
- Tian W, Guner G, Miyamoto H, et al. Utility of uroplakin II expression as a marker of urothelial carcinoma. Hum Pathol. 2015;46(1):58-64.
- Zhao G, Wang C, Tang Y, et al. Glandular differentiation in pT1 urothelial carcinoma of bladder predicts poor prognosis. Sci Rep. 2019;9(1):5323.