CASE IV:
Signalment:
19 Year old, female, African Green Monkey (Chlorocebus aethiops sabaeus).
History:
A 19-year-old, multiparous, fourth-generation colony-bred female African green monkey (Chlorocebus aethiops sabaeus) developed a swelling in the lateral aspect of the right thigh region over the course of 5 years. The animal was maintained exclusively for breeding and was not involved in any experimental procedures.
While housed in an outdoor breeding group, the monkey was observed by caretakers to be limping and unable to bear weight on her right hind limb. Upon closer inspection, the lateral thigh region appeared swollen and bruised.
The monkey was treated with multiple doses of nonsteroidal anti-inflammatory drugs and dexamethasone with only partial regression of the swelling. On palpation, the lateral thigh was firm and warm, with no additional abnormalities noted on physical examination.
Gross Pathology:
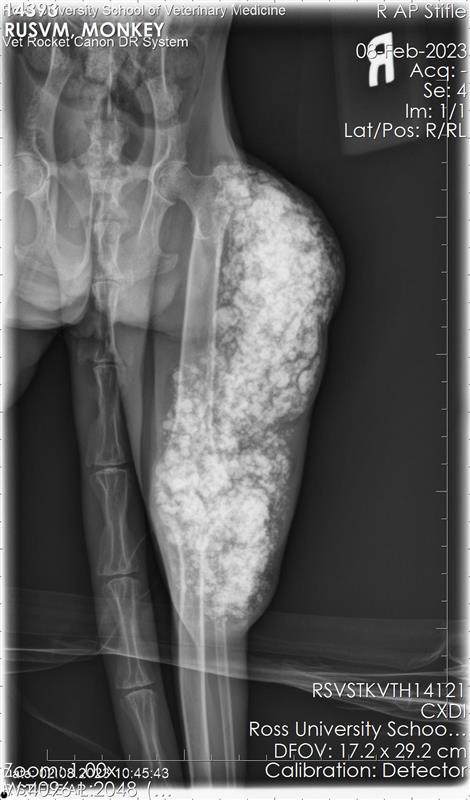
On postmortem examination, the carcass weighed 3.83 kg and had a body condition of 2 out of 5. The swollen area on the right pelvic limb exhibited mild alopecia and crusting. Postmortem radiographs showed an extensive, multilocular radiopaque mass within soft tissues surrounding the latero-caudal
aspect of the right hip and stifle, extending from the level of the greater trochanter to approximately 10 cm distal to the tibiofemoral joint. The mass did not invade long bones.
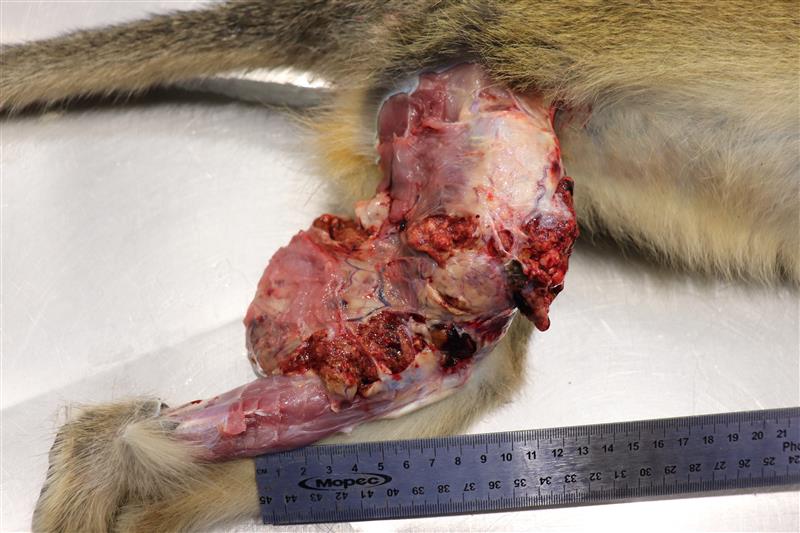
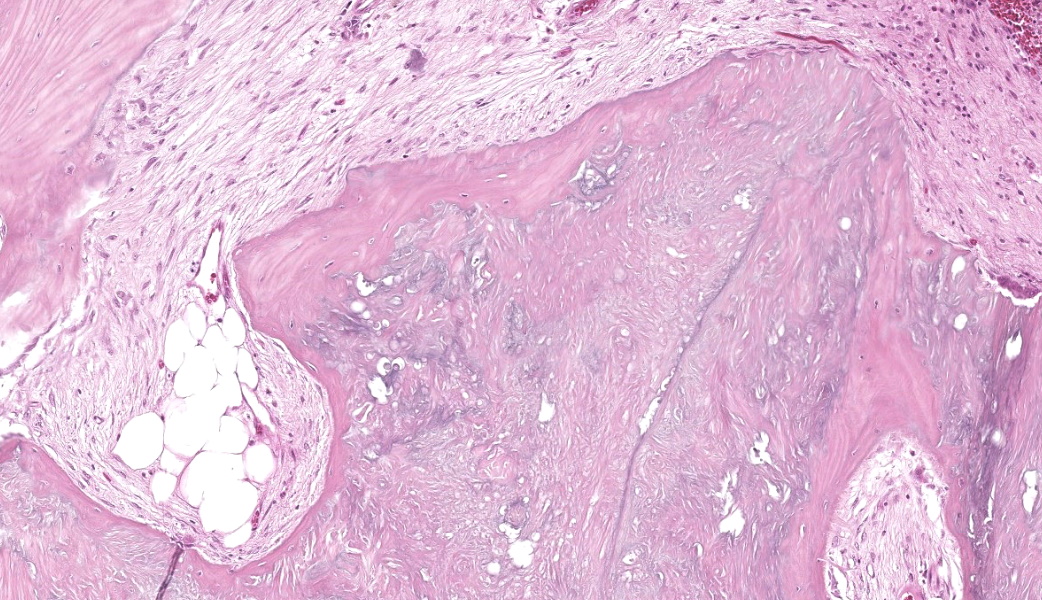
Dissection revealed a multilobulated, non-encapsulated, crepitant, yellow to red mass measuring 11 x 5 x 4 cm, infiltrating and replacing the subcutaneous tissue and skeletal muscles of the right thigh.
The mass contained numerous hard, white spicules, suggesting mineralization. The periosteum of the right femur was focally and extensively irregular. No gross pathological alterations were observed in the internal organs.
Laboratory Results:
Laboratory data was not available for this case.
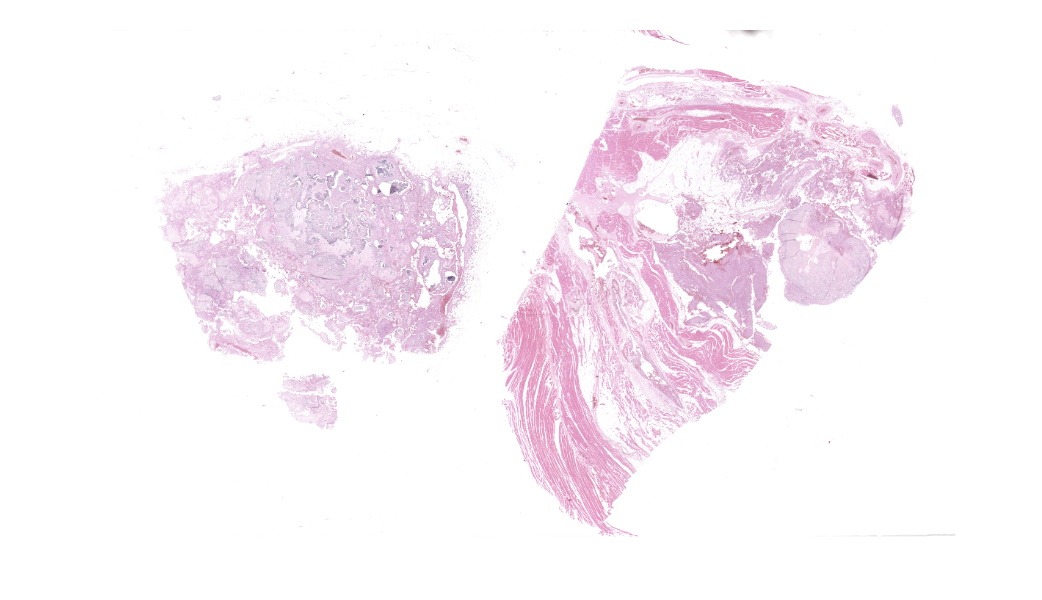
Microscopic Description:
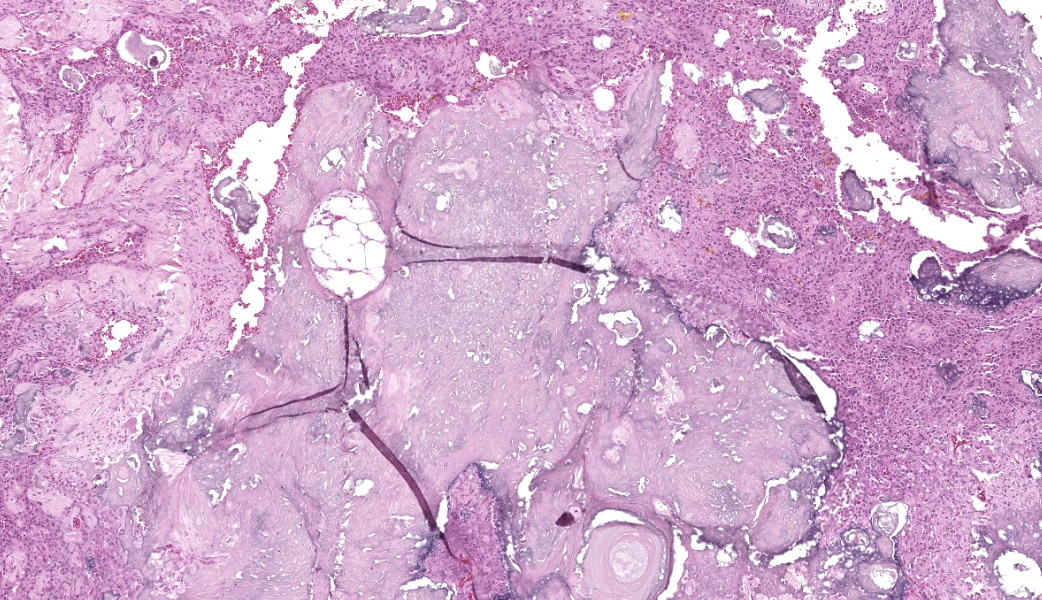
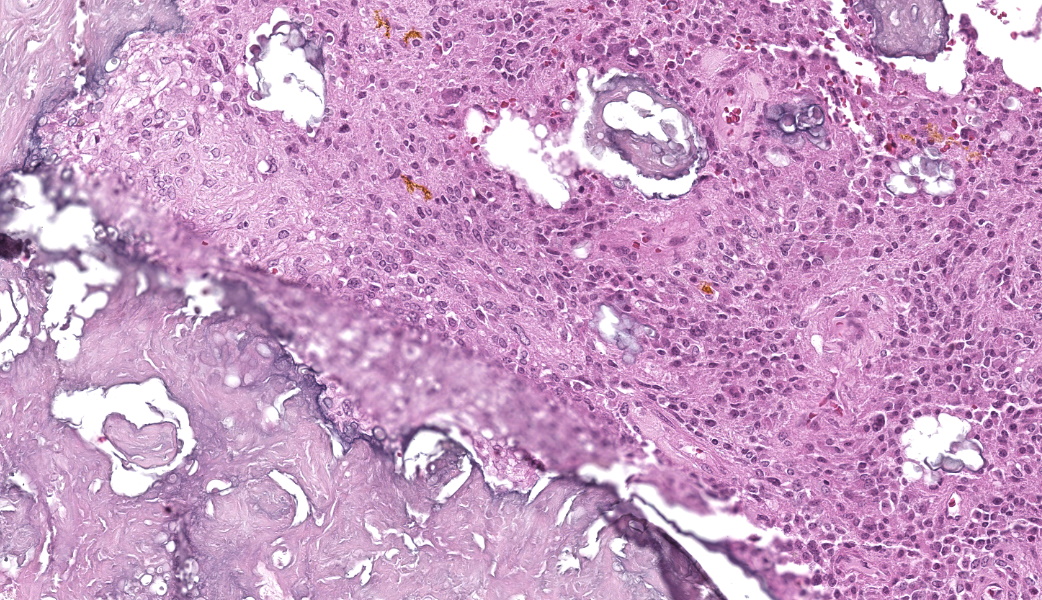
The affected skeletal muscle and subcutis were infiltrated and expanded by abundant, irregular, globoid amphophilic to basophilic material, consistent with mineralization. This mineralized material was surrounded by extensive fibrous connective tissue proliferation (fibrosis), admixed with numerous macrophages, lymphocytes, and fewer multinucleated giant cells containing phagocytosed mineral material. Scattered throughout the mass, rare foci of osteoid formation were observed, characterized by a low number of empty lacunae and necrotic osteoids undergoing remodeling. Multifocally within the mineralized soft tissue lesions, multiple tortuous blood vessels with thickened tunica media were observed. Additionally, there were areas of extensive hemorrhage, neovascularization, and granulation tissue formation infiltrating and replacing the surrounding adipose tissue.
Contributing Institution:
Pathology Department, Ross University School Of Veterinary Medicine Ross University School Of Veterinary Medicine
Contributor's Morphologic Diagnosis:
Skeletal muscle: Granulomatous myositis and steatitis with soft tissue mineralization, chronic, focally extensive, severe, with fibrosis and rare osseous metaplasia.
Contributor's Comment:
Soft tissue mineralization is broadly categorized into three types based on underlying pathophysiologic mechanisms: dystrophic, metastatic, and idiopathic calcification.5,7
Dystrophic calcification is characterized by the deposition of mineral in areas of tissue injury, occurring despite normal serum levels of calcium and phosphate. It is often associated with prior injury, necrosis, inflammation, or neoplasia, and has been reported following chronic inflammatory conditions. Metastatic calcification occurs primarily in humans and is associated with disturbances in calcium or phosphate metabolism, most commonly hypercalcemia, hyperphosphatemia or a combination of both. It is typically observed in conditions such as chronic renal failure, end-stage kidney disease, or vitamin D toxicosis. Idiopathic calcification occurs without identifiable tissue injury or underlying metabolic abnormality. No specific cause is evident, although breed or familial predisposition may be present.5,7
In humans, Hyperphosphatemic Familial Tumoral Calcinosis (HFTC) results from either a deficiency of intact FGF23 or impaired FGF23 signaling, most ofteninherited in an autosomal recessive pattern. Mutations in GALNT3, FGF23, KL(encoding KLOTHO), as well as the presence of autoantibodies targeting FGF23, disrupt FGF23 production or activity. This disruption results in hyperphosphatemia with high-normal calcium levels, leading to an elevated calcium-phosphate product, which is believed to play a key role in the development of ectopic calcification.1
In animals, gross lesions associated with calcinosis circumscripta have been documented in a variety of anatomical locations, including the tongue, margins of the pinnae, spine, salivary glands, aorta, mammary glands, small intestine, footpads, paravertebral soft tissues, cheeks, and both right and left hind limbs.4,5,7
In dogs, cats, and horses, the lesions typically present as bulging, fluctuant, or cystic masses that may be variably ulcerated and contain chalky white mineralized material.4
In a study comprising 77 canine cases, lesion diameters ranged from 2 to 13 cm, with the majority measuring between 0.5 and 3 cm. Lesions were solitary in 82% of cases and multiple in 18%, with no apparent symmetry in the latter. Grossly, lesions varied from small, firm, white nodules to large cystic masses filled with abundant chalky white material. Some lesions were freely movable, whereas others were firmly adherent to the surrounding tissues. On cut surface, most lesions exhibited multifocal nodules containing gritty, chalky white material.7
In a documented case involving a cynomolgus macaque (Macaca fascicularis), multiple raised, nodular masses of variable size were observed on both, the left and right feet. On cross-section the lesions consisted of firm subcutaneous nodules containing white, chalk-like material, and no gross pathological abnormalities were identified in the internal organs.5
Histologically, calcinosis circumscripta is characterized by aggregates of amorphous to granular, lightly to deeply basophilic mineralized material. These deposits are typically surrounded by macrophages, multinucleated giant cells, and occasional lymphocytes, and are delineated by fibrous connective tissue bands. As the lesion progresses, mineralization becomes more extensive and is accompanied by pronounced fibrosis. The associated inflammatory response may diminish over time, and areas of osseous or cartilaginous metaplasia may develop. In certain cases, epidermal sequestration or transepidermal elimination of mineralized material may lead to ulceration.2,3,4
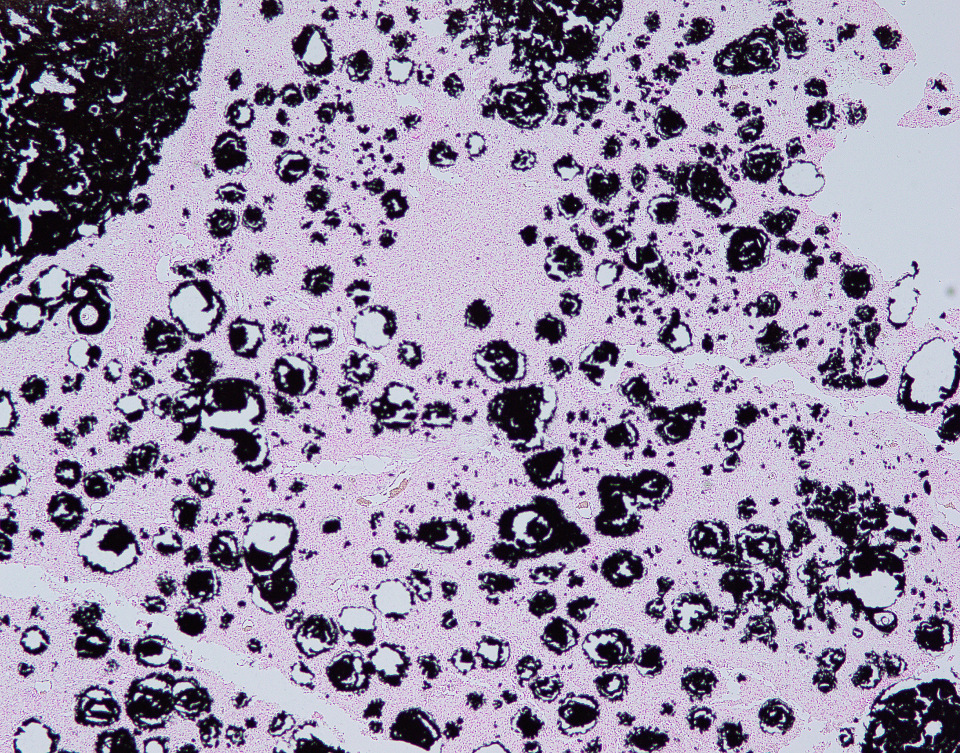
In this case, the lesions were confined to the right pelvic limb, with no evidence of involvement of other organ systems. A diagnosis of calcinosis circumscripta (tumoral calcinosis) was established based on the morphological, histopathological and immunohistochemical characteristics of the lesion. Mason trichrome, Von Kossa and Perl's Prussian blue stains were performed and were positive for the fibrous connective tissue (the first one) and the globular amorphous mineral (the last two). The presence of iron within the mineralized material is intriguing. Anecdotally, the African Green Monkeys on this facility tend to present hepatic iron overload and multifocal organ hemosiderosis. Extraskeletal osteosarcoma was considered as a potential differential. However, the cellular infiltrate was immunopositive for IBA-1 (histiocytes/macrophages) and immunonegative for SATB2 (osteoblastic origin, osteosarcoma), alpha-SMA38 (mature myofibroblast, smooth muscle tumor), SOX10 (neural crest origin, melanoma and certain soft tissue neoplasms), and S100 (amelanotic melanoma and certain soft tissue neoplasms). Based on clinical history of this animal, the lesion in this case most likely represents dystrophic calcification secondary to prior traumatic injury, as has been described in other nonhuman primates. To date, neither metastatic nor idiopathic calcification has been reported in this species.
JPC Morphologic Diagnosis:
Subcutaneous fat and skeletal muscle: Mineralization, chronic, focally extensive, severe, with granulomatous inflammation and osseous metaplasia.
JPC Comment:
The lesions in this fascinating case, while most consistent with calcinosis circumscripta, also shared histologic overlap with a condition known as myositis ossificans (also known as myositis ossificans traumatica). Myositis ossificans (MO) presents as a focal mass in the skeletal muscle, most commonly associated with the limbs, such as the biceps or thigh muscles.8 The pathogenesis of MO is speculative, but is believed to occur due to trauma and the resulting granulomatous inflammation, fibrosis, mineralization, and ossification that occur over months. Myositis ossificans is rarely reported in veterinary species, with several case reports in dogs, horses, cats, and a vampire bat.3
There is substantial histologic overlap between MO and calcinosis circumscripta; both lesions can have a mass effect due to mineral, fibrosis, granulomatous inflammation, and, occasionally, bone. Calcinosis circumscripta typically has a larger proportion of mineral and tends to occur in the skin and subcutis, often over pressure points. In contrast, MO has a variable amount of mineral versus bone depending on the stage of the lesion and occurs primarily within the skeletal muscle.2 In this case, participants agreed that there were features of both present, but could not land squarely on one or the other. It is possible that the chronicity of this lesion represents a continuum between these two in this particular case.
Participants reviewed the three main types of pathologic calcification (dystrophic, metastatic, and idiopathic) and how each relates to systemic calcium status. Dystrophic calcification occurs in necrotic or damaged tissues and is typically associated with normal serum calcium.6 Metastatic calcification, by contrast, reflects hypercalcemia, and preferentially affects tissues involved in acid-base exchange, including the gastric lamina propria, kidneys, lungs, and blood vessels.6 Finally, idiopathic calcification is caused by unknown metabolic triggers, but is associated with excess endogenous or exogenous corticosteroids in both veterinary and human medicine.6
A valuable discussion was held on the functional differences between M1 and M2 macrophages and the role of CD4+ T-helper cells in driving this differentiation. M1 macrophages, associated with pro-inflammatory responses, express CD68 and CD80, whereas M2 macrophages, associated with tissue repair and remodeling, express CD163 and CD206. Dr. Alves? lab ran these markers for this case, and the immunophenotype confirmed a predominance of M2 macrophages within the lesion. This finding aligned well with the chronicity of the process and the extensive remodeling present.
Overall, this case served as an excellent review of the mechanisms and morphologic patterns of tissue mineralization, the role of macrophage differentiation in chronic lesions, and the importance of integrating lesion distribution, chronicity, and tissue context in cases of mineralization.
References:
- Boyce AM, Lee AE, Roszko KL, Gafni RI. Hyperphosphatemic Tumoral Calcinosis: Pathogenesis, Clinical Presentation, and Challenges in Management. Front Endocrinol (Lausanne). 2020;11:293.
- De Paolo M, Gracis M, Lacava G, Vapniarsky N, Arzi B. Management of bilateral pterygoid myositis ossificans-like lesion in dogs. Front Vet Sci. 2022 Oct 10;9:992728.
- Hausmann JC, Manasse J, Churgin S, Steinberg H, Clyde VL, Wallace R. Myositis ossificans traumatica in a vampire bat (Desmodus rotundus). J Zoo Wildl Med. 2016 Sep;47(3):895-899.
- Mauldin EA, Peters-Kennedy J. Integumentary system. In: Maxie MG, ed. Jubb, Kennedy & Palmer's Pathology of Domestic Animals. Vol 1. 6th ed. St. Louis: Elsevier. 2016: 509?736.
- Radi ZA, Sato K. Bilateral dystrophic calcinosis circumscripta in a cynomolgus macaque (Macaca fascicularis). Toxicol Pathol. 2010;38(4):637-641.
- Sakals S, Peta HG, Fernandez NJ, Allen AL. Determining the cause of hypercalcemia in a dog. Can Vet J. 2006;47(8):819-821.
- Tafti AK, Hanna P, Bourque AC. Calcinosis circumscripta in the dog: a retrospective pathological study. J Vet Med A Physiol Pathol Clin Med. 2005;52(1):13-7.
- Vilar JM, Ramirez G, Spinella G, Martinez A. Kinematic characteristics of myositis ossificans of the semimembranosus muscle in a dog. Can Vet J. 2010 Mar;51(3):289-92.