Conference 18, Case 2
Signalment:
7-week-old, intact male, mixed breed dog (Canis lupus familiaris)
History:
This puppy came from a litter of 14 born to a rescued, unvaccinated dam. Half of the litter failed to thrive and was reportedly unable to latch; affected puppies were subsequently tube fed. There was concern for aspiration pneumonia and antibiotic treatment was initiated. The puppies had been dewormed and received the first of a vaccine series. This puppy was acutely anorexic, and was bottle fed prior to death.
Gross Pathology:
The liver was diffusely mottled yellow to red and had tens of pinpoint, flat, red discoloration randomly scattered across the capsular surface. The lungs were diffusely mottled tan to pink, slightly rubbery, and exuded a moderate amount of clear to yellow, watery to slightly viscous fluid on cut section. On cut surface of the brain, primarily within the thalamus, was a fairly well demarcated region of red to brown discoloration and there were pinpoint regions of red-brown discoloration throughout the adjacent parenchyma. p
Laboratory Results:
- Canine adenovirus-2 PCR: Positive
- Aerobic culture, lung: No growth
- Aerobic culture, liver: No growth
- Canine herpesvirus PCR: Negative
Microscopic Description:
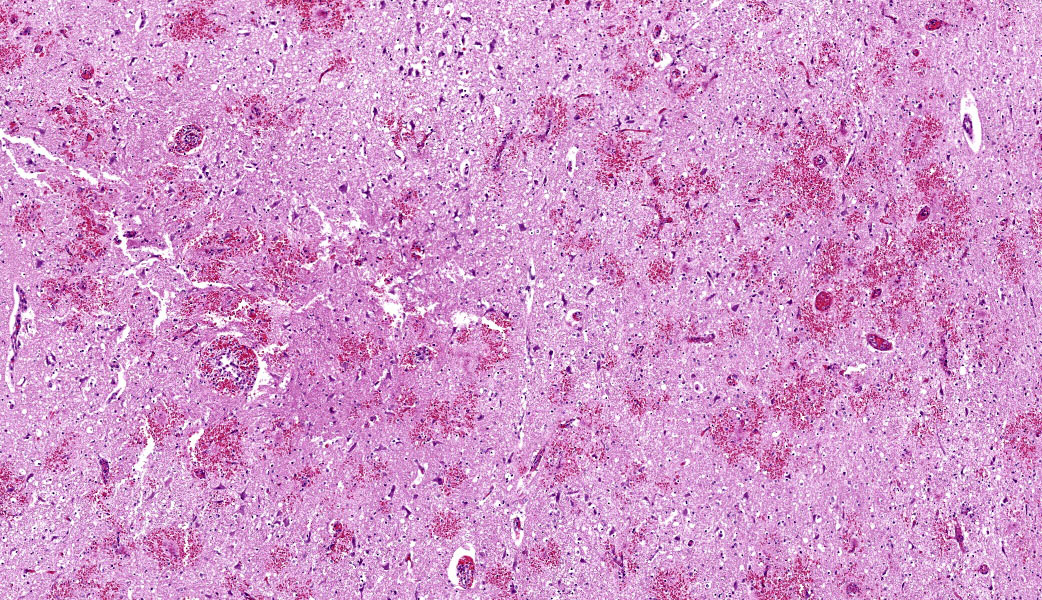
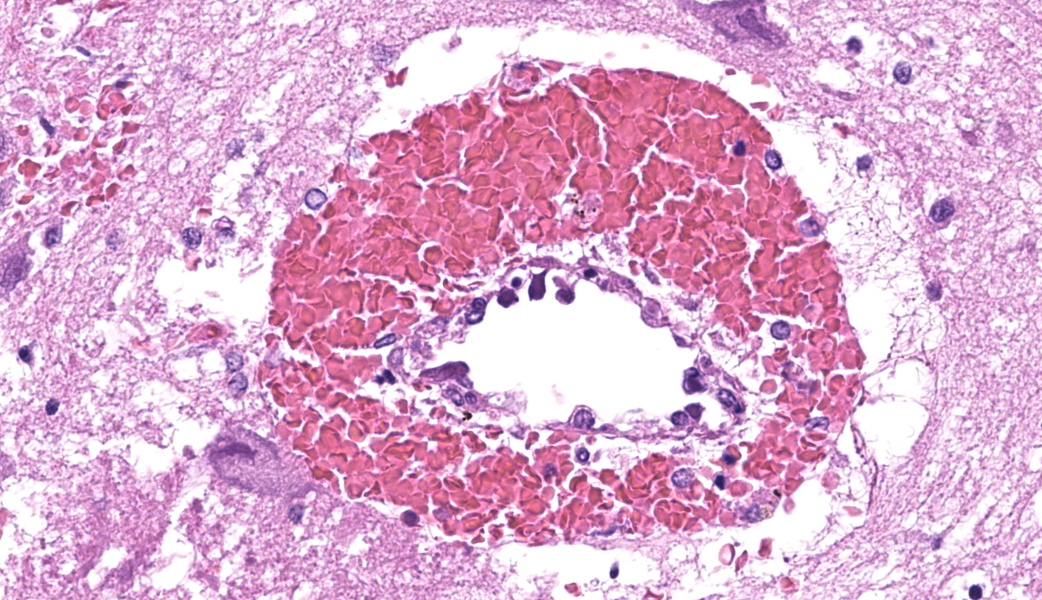
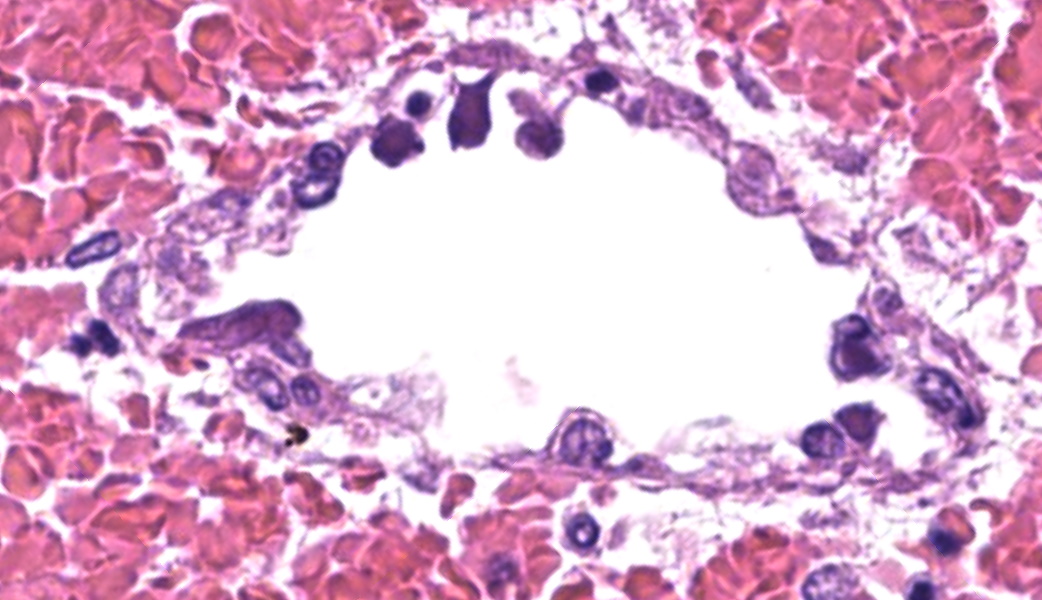
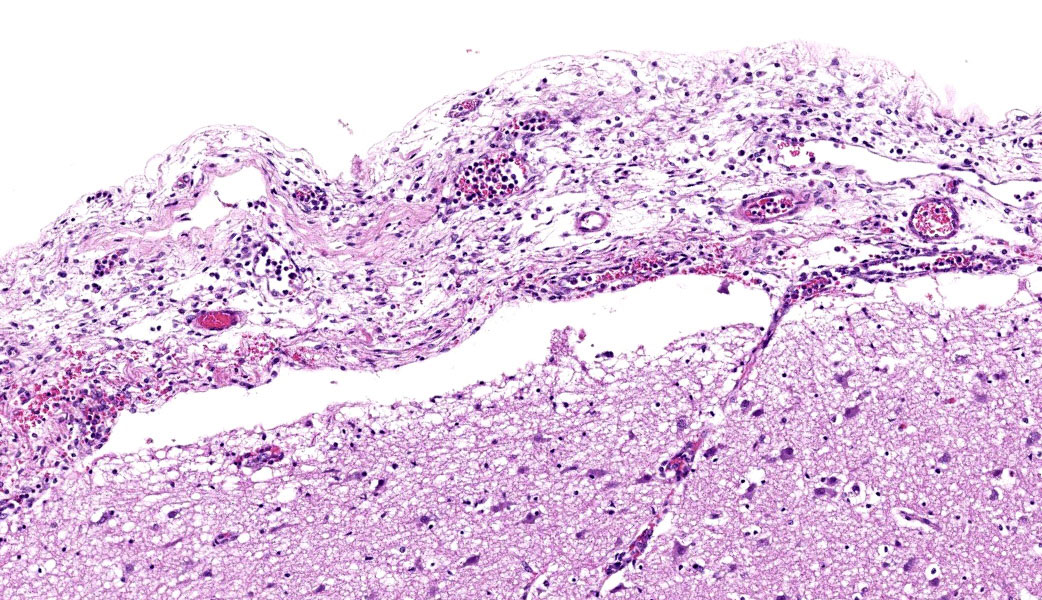
Throughout the cerebral gray matter, small caliber vessel walls were frequently obscured by mild to moderate amounts of eosinophilic, fibrillar, proteinaceous material and hemorrhage. Endothelial cells frequently contained large, eosinophilic, intranuclear inclusions which peripheralize chromatin. Affected vessels were frequently cuffed by moderate numbers of macrophages and lymphocytes, and there were frequent, small areas of hemorrhage spilling into the adjacent tissue. The meninges overlying the cerebellum were moderately expanded by lymphocytes and macrophages, and endothelial cells frequently contained intranuclear inclusions similar to those found in the cerebral vessels.
Contributor’s Morphologic Diagnoses:
Severe, multifocal, acute, histiocytic vasculitis and meningitis with cerebral hemorrhage and intranuclear viral inclusions.
Contributor’s Comment:
Adenoviruses are linear, double-stranded DNA, icosahedral viruses which are subdivided into four genera – Mastadenovirus, Aviadenovirus, Atadenovirus, and Siadenovirus. Mammalian adenoviruses generally belong to the Mastadenovirus genus and are categorized based on neutralization assays. This viral family replicates within nuclei, leading to the formation of characteristic intranuclear inclusions seen in a variety of cell types depending on the species and site of infection. Canine adenovirus-1 infection, colloquially known as infectious canine hepatitis, most often infects hepatocytes leading to acute hepatitis, but can infect the endothelium, causing disseminated intravascular coagulation and hemorrhage. Infection with canine adenovirus-2 is typically localized to the respiratory system, causing mild, non-specific signs of acute respiratory disease which eventually resolve.8
Although unusual, systemic infection with canine adenovirus-2 has previously been reported.1 In one report, neurologic signs were noted in 3 of 4 puppies out of a litter of 9 who were diagnosed with CAV-2 systemic infection. Two of these puppies displayed concurrent, mild respiratory signs. Interestingly, as in the submitted case, these puppies were born to an unvaccinated dam. In contrast to the submitted case, despite the clinical presence of neurologic abnormalities prior to death, nervous system lesions were not present histologically, although virus was detected via PCR in the brain in one affected animal.1
While reports of CAV-2 induced vasculitis are sparse, other species of adenovirus-associated vasculitis are more common. Perhaps the most similar syndrome of adenovirus-associated vasculitis is seen in adenovirus hemorrhagic disease of cervids. Clinically, this disease presents similarly to epizootic hemorrhagic disease, with histologic lesions including systemic vasculitis and petechiae, along with intranuclear inclusions which separate this disease from epizootic hemorrhagic disease. Affected animals have numerous intranuclear inclusion bodies found in the endothelial cells of a variety of organs, including the brain, which are occasionally associated with fibrin thrombi or fibrinoid necrosis, vasculitis, and/or perivascular cuffs.2,9 In affected animals, lesions are most frequent in the lungsand brain, although hepatic lesions may also be seen, as in this case.9
Human adenovirus of an undetermined serotype has been reported as a cause of acute retinal necrosis due to occlusive vasculitis.7 One study examining the presence of vasculitis associated with severe respiratory infections found uncommon reports of thrombosis or vasculitis associated with human respiratory adenovirus infections.5 Following a variety of immunosuppressive therapies and stem cell transplant, adenovirus-associated vasculitis with microscopic regions of cortical necrosis and hemorrhage were seen in a human multiple myeloma patient. In this case, abundant viral inclusions were found in the endothelial cells, but not epithelial cells within the examined organs.4
In the current case, CAV-2 was detected via PCR and immunohistochemistry within the histologically observed cerebral and meningeal lesions associated with vasculitis. Previously reported cases of disseminated CAV-2 infection with vasculitis have been proposed to be associated with other infectious agents or diseases causing immunosuppression. There was not evidence of an underlying cause for immunosuppression in the current case, and there was no evidence of concurrent systemic disease. Thus, the vascular lesions associated with CAV-2 appear to be the primary cause of disease in this puppy. As in the above discussed previously reported case of CAV-2 infections associated with neurologic signs in a litter of puppies, it appears likely that the lack of maternal vaccination may have contributed to disease in this case.1
Contributing Institution:
National Institute of Health Comparative Biomedical Scientist Training Program
JPC Diagnosis:
Cerebrum: Vasculitis, neutrophilic and lymphocytic, subacute, multifocal to coalescing, marked, with fibrin thrombi, lymphohistiocytic meningitis, and endothelial intranuclear viral inclusions.
JPC Comment:
This case stimulated great discussion during conference and the WSC team would like to thank this contributor for sending in such an interesting submission and a fantastic write-up! MAJ Sulkosky stated that, although the causative agent was diagnosed as CAV-2 by the contributor, this case should be considered classic for a CAV-1 infection in a puppy. CAV-1 has been seen a couple of times recently in the WSC in 2022, Conference 22, Case 3, and in 2019, Conference 16, Case 1.
Conference participants speculated as to why CAV-2 was isolated in this case, as there was no mention of testing for CAV-1, and wondered if there had potentially been cross-reactivity on the test. The papers that describe CAV-2 encephalitic disease in dogs lack supporting evidence to demonstrate that the lesions were caused incontrovertibly by CAV-2, so attendees remained unconvinced.1,3 Most wondered if this case was instead a CAV-1 encephalitis and the CAV-2 positivity was either a false positive, was showing as positive due to the recent history of a distemper combo vaccine in this puppy, or was due to a co-infection with CAV-1 that was just not tested for. It is still unclear if CAV-2 can truly cause vasculitis in the brains of puppies. CAV-2 is considered primarily a mild respiratory pathogen, and there are very rare reports of other organs affected.p
There is one paper that describes 2 cases of fatal encephalitic CAV-1 infection in puppies, but the sequence of the pVII gene in that CAV-1 was more closely related to that seen in CAV-2, suggesting a possible recombinant strain.10 The pVII gene in canine adenoviruses encodes an arginine-rich core protein (pre-VII) which is cleaved to form pVII. The pVII protein is responsible for condensing the viral DNA within the capsid, acting like a histone protein.6 pVII plays a critical role in the virus's structure and, due to its nuclear localization, serves as a target for labeling in research studies.6
Wrapping up this spirited case discussion was a review of the viruses that form paracrystalline arrays that can be seen with electron microscopy. This list of viruses can be remembered using the acronym “PPPICCA,” which stands for paramyxovirus, picornavirus, polyomavirus, iridovirus, circovirus, calicivirus, and adenovirus.
References:
- Benetka V, Weissenböck H, Kudielka I, Pallan C, Rothmüller G, Möstl K. Canine adenovirus type 2 infection in four puppies with neurological signs. 2006;158(3):91–94.
- Burek-Huntington K, Miller MM, Beckmen K. Adenovirus hemorrhagic disease in Moose (Alces americanus gigas) in Alaska, USA. J Wildl Dis. 2021;57(2):418–422.
- Chander V, et al. Isolation and genetic characterization of canine adenovirus type 2 from a domestic dog showing neurological symptoms. Braz J Microbiol. 2021 Dec;52(4):2521-2528.
- Claveau JS, LeBlanc R, Ahmad I, et al. Cerebral adenovirus endotheliitis presenting as posterior reversible encephalopathy syndrome after allogeneic stem cell transplantation. Bone Marrow Transplantation. 2017:52;1457–1459.
- Dolby HW, Potey P, Wilder-Smith AB, et al. Histological evidence of pulmonary microthrombosis and vasculitis in life-threatening respiratory virus diseases. Open Forum Infect Dis. 2021;8(2).
- Morrison MD, Onions DE, Nicolson L. Complete DNA sequence of canine adenovirus type 1. J Gen Virol. 1997;78(4):873-878.
- Özdemir HB, Özdal PÇ. Human adenovirus: An unusual causative agent for acute retinal necrosis presented with central retinal vascular occlusion. Ocul Immunol Inflamm. 2020;28(5):749–753.
- Quinn PJ, Markey BK, Leonard FC, FitzPatrick ES, Fanning S, Hartigan PJ. Adenoviridae. In: Veterinary Microbiology and Microbial Disease, 2nd ed. Blackwell Publishing; 2011:588-592.
- Woods LW, Swift PK, Barr BC, et al. Systemic Adenovirus Infection Associated with High Mortality in Mule Deer (Odocoileus hemionus) in California. Vet Pathol. 1996;33.
- Wong M, Woolford L, Hasan NH, Hemmatzadeh F. A Novel Recombinant Canine Adenovirus Type 1 Detected from Acute Lethal Cases of Infectious Canine Hepatitis. Viral Immunol. 2017;30(4):258-263.