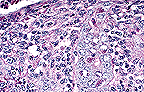
 Necrotizing colitis with intraepithelial
coccidial schizonts in a calf. (40X, HE, 110K)
Necrotizing colitis with intraepithelial
coccidial schizonts in a calf. (40X, HE, 110K) Necrotizing colitis with intraepithelial
coccidial schizonts in a calf. (40X, HE, 110K)
Necrotizing colitis with intraepithelial
coccidial schizonts in a calf. (40X, HE, 110K)
 Iintraepithelial coccidial
schizonts in the colonic epithelium of a calf. (100X, HE, 114K)
Iintraepithelial coccidial
schizonts in the colonic epithelium of a calf. (100X, HE, 114K)
Signalment: 4-week-old male crossbred Hereford calf
History: The calf was submitted to the referring veterinarian with the complaint of diarrhea. Clinical examination revealed that the calf was thin and severely dehydrated. The perineum was pasted with feces containing some flecks of blood. Fecal examination for coccidia was negative. Treatment consisted of oral and parental fluids, antibiotics, and an antispasmodic. The calf died 24 hours after initiation of treatment.
Gross Pathology: History indicated that the calf was emaciated; serous atrophy of body fat depots was noted. The intestinal tract was devoid of contents except for a small quantity of bloody feces in the cecum and colon. Formalin-fixed sections of jejunum, ileum, and colon were submitted for histological examination.
Laboratory Results: None submitted
Contributor's Diagnosis and Comments: Colitis, fibrinopurulent, diffuse, severe, with intra-epithelial coccidia.
Microscopic lesions vary in appearance among the histological slides. In the section of colon examined, clumps of fibrin containing cellular debris and inflammatory cells are adhered to the mucosa. Most crypt epithelial cells contain schizonts, gamonts, or developing oocysts. Many intestinal crypts are distorted or have been destroyed as a result of coccidial infection. Erosion and ulceration of the mucosa have occurred. A lymphoplasmacytic inflammatory reaction is present in the lamina propria.
The most likely etiologic agent is Eimeria zuernii which is one of the most common and pathogenic coccidia of ruminants. The other is E. bovis. Life cycles of both agents are similar. Large schizonts typical of either E. bovis or E. zuernii were not observed in multiple sections of jejunum and ileum examined. Eimeria bovis infects endothelial cells in lacteals of the jejunum located several meters anterior to the ileocecal valve; whereas, E. zuernii schizonts are found in the lamina propria of the terminal ileum. Secondary schizonts and gamonts of E. zuernii develop in the glands of the cecum and colon but not terminal ileum.
Coccidiosis is characterized by diarrhea which can progress to dysentery and death. Morbidity can be high but mortality is generally low. Infected animals become dehydrated, hyponatremic, and anemic. The duration of disease is usually 3 to 10 days, after which most cases recover due to the self-limiting nature of disease. In heavily infected animals, death may occur before substantial numbers of oocysts are shed in the feces which may explain the failure to demonstrate oocysts in the feces of this calf.
AFIP Diagnosis: Colon: Colitis, necrotizing, subacute, diffuse, moderate, with erosions, and myriad intracellular coccidia, Hereford-cross, bovine, etiology consistent with Eimeria sp.
Conference Note: The life cycles of Eimeria zuernii and Eimeria bovis are similar. They require two generations of schizogony before gametogony occurs. Gametogony preceeds oocyst formation, which peaks on the 19th to 20th day post-infection. A 30-day-old animal may not have lived long enough for the coccidia to complete their life cycle, explaining the low numbers of oocysts present in the tissue section and the failure to find them on fecal floatation.
Oocysts can also be utilized to differentiate between many coccidian genera. Eimeria oocysts contain 4 sporocysts with 8 sporozoites (2 per sporocyst), while Isospora oocysts contains 2 sporocysts with 8 sporozoites (4 per sporocyst).
Conference participants discussed various causes of metabolic acidosis. Death in animals infected with Eimeria often occurs as the result of metabolic acidosis related to diarrhea. Understanding the physiology of acid-base imbalance is useful in determining the cause and implementing proper therapy. There are two mechanisms of metabolic acidosis, secretory and titrational. Titrational metabolic acidosis occurs when organic acids accumulate. Clinically important organic acids include lactic acid in shock and grain overload, ketone bodies in diabetes and starvation in ruminants, uremic acids in renal failure, and organic poisons. The electrolyte pattern in titrational acidosis is low plasma HCO3-, normal serum Cl-, and a high anion gap [(Na+ + K+) - (Cl- + HCO3-)].
Secretory acidosis occurs when bicarbonate is excreted from the body in bicarbonate-rich fluids such as saliva, urine (rich in bicarbonate in cases of renal tubular acidosis), and gastrointestinal secretions lost in diarrhea, as occurred in this case. The electrolyte pattern expected in secretory acidosis is low plasma HCO3-, high serum Cl-, and a normal anion gap.
Contributor: Veterinary Diagnostic Center, University of Nebraska-Lincoln, Lincoln, NE 68583-0907.
References:
International Veterinary Pathology Slide Bank: Laser disc frame #4049, 4839, 5062, 9692, 9693, 22777
 Protozoal cyst of T. cruzi
in the heart of a puppy. (40X, HE, 120K)
Protozoal cyst of T. cruzi
in the heart of a puppy. (40X, HE, 120K)
 Amastigotes of T. cruziin
the myocarium of a puppy. (100X, HE, 118K)
Amastigotes of T. cruziin
the myocarium of a puppy. (100X, HE, 118K)
History: Multiple fixed tissues from a female 13-week-old English Pointer puppy from south Texas were received for evaluation. No history accompanied the submission.
Gross Pathology: None submitted.
Laboratory Results: None submitted.
Contributor's Diagnosis and Comments: Heart: Pancarditis, lymphoplasmacytic and granulomatous, diffuse, chronic, severe, with multifocal cardiomyofiber necrosis, and intrasarcoplasmic protozoal amastigotes, English Pointer, canine.
Diffusely in the endocardium and in the interstitium of the myocardium, there is marked inflammation composed of plasma cells, lymphocytes, macrophages, and lesser numbers of neutrophils, accompanied by moderate perimysial fibrosis. This inflammation widely separates, and multifocally replaces, myocardial fibers. Multifocally, myocardial fiber diameter is decreased, and occasionally fibers are eosinophilic, fragmented, and lack striations. Scattered fibers contain pseudocysts which contain multiple amastigotes, each of which has a basophilic kinetoplast and a nucleus.
Pulmonary edema, and marked centrilobular congestion in the liver were also noted. The kidney, pancreas, ileum, colon, small intestine, stomach, and spleen, contained no significant lesions.
Trypansoma cruzi is a protozoon that causes Chagas disease in humans, a disease that is endemic in Central and South America. It is transmitted by insects of the Reduviidae family ("kissing bugs"). The insects become infected by ingesting infected blood. The trypanosomes transform and multiply in the insect's gastrointestinal tract. In South America, transmission to mammals is by the deposition of infective insect feces on a bite wound inflicted by the feeding insect. In the United States, T. cruzi has been reported in 18 species of mammals. Reports in mammals come from all the southern states as well as Maryland. The most common clinical presentation in dogs is acute myocarditis. Chronic dilatative cardiomyopathy has also been reported. It appears that hunting dogs are at increased risk of infection. The reduviid vectors in North America are Triatoma sanuisuga and Triatoma protracta. Since the North American vectors defecate long after taking a blood meal, two additional modes of transmission have been postulated in the US, namely the ingestion of the infected vectors by the dog, and transmission by eating or fighting with wild hosts such as opossums and raccoons.
Differentials for this protozoan might include Toxoplasma gondii and Leishmania donovani. The trophozoites of T. gondii however do not contain a kinetoplast, and in addition their pseudocysts are weakly periodic acid-Schiff positive. Amastigotes of L. donovani do contain a kinetoplast; however, differentiation from T. cruzi can generally be made on the basis of the organs affected.
AFIP Diagnosis: Heart: Pancarditis, necrotizing, subacute, diffuse, severe, with intrasarcoplasmic amastigotes, English Pointer, canine, etiology consistent with Trypanosoma cruzi.
Conference Note: The classic life cycle forTrypanosoma cruzi utilizes reduviid insects as a vector. When these insects feed on infected individuals, they also ingest trypomastigotes (trypanosomes) of T. cruzi. The trypomastoigotes localize in the midgut of the reduviid bug and multiply and differentiate from transient amastigotes (leishmanial forms) to epimastigotes (crithidial forms), and finally to metacyclic trypomastigotes. The latter accumulate in the rectum and are discharged with the feces, sometimes onto a potential host. Infection takes place by fecal contamination of mucous membranes or abraded skin or by ingestion of the insect. In the mammalian host, the metacyclic trypomastigotes enter histiocytes and proliferate in the amastigote form at the local site where a fibrous encapsulation may obstruct lymphatics causing localized edema. Amastigotes pass to local lymph nodes and then disseminate throughout the body via the lymphatic system. Liver, lungs, spleen, bone marrow, cardiac muscle and cerebral cortex are affected. In these sites, amastigotes proliferate, causing rupture of host cells and release of trypomastigote forms into the blood. Trypomastigotes penetrate various types of cells including cardiac, smooth, and skeletal muscle; the nervous system, skin, gonads, intestine, bone marrow, and placenta may also be infected. Intracellular amastigotes undergo repeated divisions to form large numbers of parasites, producing the parasitic cyst. After a few days, the amastigotes retransform into trypomastigotes and burst out of the parasitic cyst. A generalized parasitemia occurs, and almost any type of tissue can be invaded, although the parasites show a preference for muscle and nerve cells. This cycle is repeated producing large numbers of amastigotes and the clinical signs and histological lesions associated with T. cruzi infection.
Trypanosoma cruzi causes an acute myocarditis that can progress to a chronic, debilitating and often fatal disease characterized by dilatative cardiomyopathy. Lesions frequently associated with chronic T. cruzi infection are pulmonary edema and hepatic congestion, due to development of congestive heart failure secondary to cardiomyocyte damage. Hepatic centrilobular congestion may result in the elevation of the liver enzymes alanine aminotransferase and aspartate aminotransferase (AST). Parasitic destruction of muscle cells, which also contain AST in significant concentrations, will contribute to serum elevations of that enzyme. Muscle damage is more accurately accessed, however, by measurements of serum levels of creatine kinase (CK) and lactate dehydrogenase (LDH).
CK is a dimeric enzyme composed of two subunits: B for brain, and M for muscle, that forms three isoenzymes (CK1-CK3). CK1 is found in nervous tissue and is not found in the serum, even during disease. CK2 is found in cardiac muscle, and CK3 is found in skeletal and cardiac muscle. CK has a half-life of 2-4 hours and returns to normal in 1 to 2 days after cesseation of injury. LDH is a tetrameric enzyme composed of two subunits, H (heart) and M (muscle). In dogs, LDH1 is the principal isoenzyme of cardiac muscle and kidney. LDH5 is the principle isoenzyme in erythrocytes and skeletal muscle. LDH has a longer half-life and will persist in the serum longer than CK or AST. These muscle enzymes may not be elevated in cases of chronic trypanosomiasis, as the cellular damage occurs long before clinical signs develop; however, they could be of diagnostic value in identifying myocardial or muscular damage during the acute form of the disease.
Contributor: Texas Veterinary Medical Diagnostic Laboratory, Drawer 3040, College Station, TX 77841.
References:
International Veterinary Pathology Slide Bank: Laser disc frame
#6950, 7210, 7244, and 11090
 Massive heaptic necrosis in
a young dog. (40X, HE, 106K)
Massive heaptic necrosis in
a young dog. (40X, HE, 106K)
Signalment: 8-month-old, mixed breed, castrated male dog.
History: The referring veterinarian diagnosed tracheobronchitis on 6/1/95. Dog was treated with trimethoprim-sulfadiazine (Tribrissen) 24mg/kg q 12 hr. Dog presented to Colorado State University with acute onset of weakness. Physical examination revealed decreased lung sounds in cranial ventral thorax and mild scleral icterus.
Gross Pathology: The liver is small (approx. 50-66% of normal size) and diffusely dark red. All hepatic lobes are pliable with sharp edges. There are multifocal, 1-2 mm, pale tan foci randomly distributed over the capsule surface and on cut surface of the parenchyma. The liver parenchyma is slightly firm and readily oozes blood from cut surfaces.
Laboratory Results:
Total bili. 2.6 (0.0-0.4)
Prothrombin 17.8 (7.5-10.5)
ALP 1021 (1.8-141)
APTT 55.4 (10.5-16.5)
ALT 2877 (10-120)
FDP >40+<80
AST 598 (16-40)
Urinalysis 2+ bilirubin
GGT 13 (0-6)
Contributor's Diagnosis and Comments: Liver: Submassive to massive hepatic necrosis, severe, diffuse, acute; with diffuse hemorrhage and dissolution of the hepatic parenchyma.
Etiology: Trimethoprim-sulfadiazine-induced hepatic necrosis.
Trimethroprim-sulfonamide (TMS) combination antibiotics are commonly used in veterinary medicine to treat a variety of infectious diseases. Although adverse reactions are infrequent, massive hepatic necrosis has been reported in dogs. Duration of therapy, type of TMS combination, and dose do not appear relevant to development of toxicity. The small number of dogs reported with TMS- associated hepatotoxicity suggests that this is an idiosyncratic reaction.
AFIP Diagnosis: 1. Liver, hepatocytes: Necrosis and loss, centrilobular
and periportal to massive, diffuse, severe, mix-breed, canine.
2. Liver: Hyperplasia, biliary, diffuse, mild, with mild subacute
periportal hepatitis and extramedullary hematopoeisis.
Conference Note: Drugs known to induce hepatic disease include mebendazole, diethycarbamazine, sodium thiacetarsamide, methoxyflurane, halothane, primidone, phenytoin, phenobarbital, mephobarbital, diazepam, phenylbutazone, and trimethoprim with sulfadiazine. Several mechanisms have been proposed to explain drug induced hepatotoxicities. Many drugs are directly toxic to hepatocytes or biliary epithelium. Other drugs may induce immune-mediated hepatic damage by reacting with host cell membranes and acting as haptens, localizing on host cell membranes and acting as antigens, or altering host proteins and inducing autoimmune reactions. Viral infections may sensitize some individuals to drugs. The mechanism of hepatocellular injury has not been determined for trimethoprim-sulfa (TMS) drugs.
Clinicopathologic findings associated with TMS therapy include reduced packed cell volume (PCV), mean cell volume (MCV) and mean cell hemoglobin (MCH). TMS inhibits sequential steps in folic acid metabolism, consequently impairing DNA synthesis which limits bacterial proliferation since absorption of folate is impaired. Most animals can absorb reduced folates and are less affected than bacteria by the the action of TMS. Large doses or long-term treatment with TMS can reduce the amount of folates available for DNA synthesis, resulting in impaired erythropoiesis and microcytic hypochromic anemia.
There are many clinical laboratory tests that can be utilized to measure hepatic injury; they fall into two general categories of tests. The first are nonfunctional tests that measure cellular enzymes which are released from damaged hepatocytes. These enzymes are alanine aminotransferase (ALT), aspartate aminotransferase (AST), sorbitol dehydrogenase (SDH), alkaline phosphatase (ALP), and gamma-gutamyltransferase (GGT). Both AST and ALT reflect changes in hepatocellular permeability (sublethal damage or necrosis); however, ALT is considered to be liver specific in the dog and cat (AST being present in erythrocytes and muscle). In the horse and bovine SDH is considered to be liver specific. Elevations of serum ALP and GGT usually indicate bile stasis.
The second category of tests measure hepatic function. Liver functions measured by clinical tests include protein synthesis (albumin, globulins, and clotting factors), bile synthesis and secretion (bilirubin and bile acids), excretion of organic anions (sulfobromophthalein and indocyanine green), ammonia and amino acid metabolism (ammonia and blood urea nitrogen) and glucose homeostasis (blood glucose). Hepatic function test values are usually altered only after hepatocellular injury involves more than half of the organ.
The laboratory results in this case indicate significant hepatocellular damage and cholestasis. Both functional and nonfunctional liver test values are greatly elevated and are consistent with the severe hepatocellular necrosis and loss seen histologically.
Contributor: Colorado State University, College of Veterinary Medicine and Biomedical Sciences, Department of Pathology, Fort Collins, CO 80523.
References:
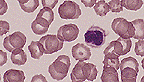 Abnormal non-lobulated neutrophil
in the peripheral blood smear of a dog. (100X, Wright's, 116K)
Abnormal non-lobulated neutrophil
in the peripheral blood smear of a dog. (100X, Wright's, 116K)
Signalment: 7-year-old intact male Basenji dog
History: CBC performed at time of endoscopic biopsy. Mucosal biopsies of small intestine consistent with immunoproliferative small intestinal disease. Dog had a history of chronic vomiting and diarrhea.
Gross Pathology: None submitted.
Laboratory Results:
WBC: 10.6 x 103/æl
HCT: 35%
NRBC: 10/100 White cells
Reticulocytes: 0.6%
RBC: 6.13 x 106/æl
Hemoglobin: 12.0 g/dl
Platelets: adequate
Plasma protein: 6.6 g/dl
BUN: 22 mg/dl
Contributor's Diagnosis and Comments: Peripheral blood smear with Pelger Huet anomaly.
Basenji dog with immunoproliferative small intestinal disease (IPSID) often have Pelger Huet anomaly.
AFIP Diagnosis: Blood smear: Granulocytic hyposegmentation, diffuse, with mature chromatin pattern and mild microcytic, hypochromic anemia, Basenji, canine.
Conference Note: Pelger-Huet anomaly is considered a benign hereditary condition that results in nuclear hyposegmentation of granulocytes, megakaryocytes, and monocytes. The condition is usually heterozygous and does not alter antimicrobial functions of neutrophils including adherence, chemotaxis, phagocytosis, and degranulation. There is no impact on the clinical health of affected dogs. The homozygous mutation is a lethal trait.
Pelger-Huet anomaly must be differentiated from pseudo-Pelger-Huet syndrome, an asynchronous neutrophil maturation associated with a left shift and chronic disease. Band cells of pseudo- Pelger-Huet have less dense chromatin patterns. Pseudo-Pelger-Huet is transient and occurs secondary to chronic infection, myeloid metaplasia, drug administration, or, in humans, neoplasia involving the bone marrow.
Many at the conference also noted changes in the RBC's, to include macrocytosis and polychromasia. The HCT is also slightly low, indicating a mild anemia. Evaluation of the Mean Cell Volume [(Hct x 10)/RBC count=57], and MCHC [(Hb concentration x 100)/Hct=34], indicate a microcytic , normochromic anemia. The absolute reticulocyte count (multiplying the RBC count by the reticulocyte percentage), was 36,780. In the dog, an absolute count of less than 80,000 reticulocytes indicates a nonregenerative response. The release of nucleated red blood cells is expected with erythroid hyperplasia; however, the degree of erythroid response in this case is inadequate.
The WBC count (Addendum) was corrected for nucleated RBC's by the following formula: (initial WBC count x 100) ö (100 + nRBC). Corrected WBC=9,630
Those attending the conference were not given the WBC count. They determined the value by counting the number of white cells in ten 40x fields innareas of the smear where RBC's just touch each other. This number (N) is inserted into the following formula: (N ö 10) x 1500= WBC count. Estimates ranged from 5700-9750.
The Basenji in this case had chronic diarrhea, anorexia, maldigestion, and alopecia and necrosis of the pinna and was diagnosed as having immunoproliferative small intestinal disease (IPSID). Basenjis with IPSID occassionally have Pelger-Huet anomaly. Immunoproliferative small intestinal disease of Basenji dogs is characterized by chronic diarrhea, malabsorption and maldigestion, intermittent anorexia, gastropathy, lymphoplasmacytic enteritis, hypergammaglobulinemia, and alopecia of the pinna and ventrum. The pathogenesis of this syndrome is not understood. A similar disease in humans is associated with abnormal IgA immunoblasts; Basenjis with IPSID often have increased levels of circulating IgA.
Some at the conference speculated that the nonregenerative anemia may have been related to the maldigestion/malabsorption syndrome diagnosed in this dog, as nutritional deficiencies (ie. iron, B vitamins) can result in erythrocytic maturation abnormalities. Anemia of chronic disorders may also play a role in this case. This type of anemia occurs in cases of chronic inflammation or neoplasia. The mechanisms are not well understood, but it appears that a combination of cytokines mediate the anemia. Inhibition of erythropoietin and impairment of iron release from macrophages are also contribututory.
Contributor: University of Miami School of Medicine, Department of Pathology (R- 46), Miami, FL 33136.
References:
International Veterinary Pathology Slide Bank: Laser disc frame #15097-15101
Dana P. Scott
Captain, VC, USA
Registry of Veterinary Pathology*
Department of Veterinary Pathology
Armed Forces Institute of Pathology
(202)782-2615; DSN: 662-2615
Internet: Scott@email.afip.osd.mil
* The American Veterinary Medical Association and the American College of Veterinary Pathologists are co-sponsors of the Registry of Veterinary Pathology. The C.L. Davis Foundation also provides substantial support for the Registry.